UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
——————————————
FORM 8-K
——————————————
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of report (Date of earliest event reported): November 10, 2025
——————————————
(Exact name of registrant as specified in its charter)
——————————————
(State or other jurisdiction of incorporation or organization) | (Commission File Number) | (I.R.S. Employer Identification No.) | ||||||||||||
(Address of principal executive offices) (Zip Code)
(858 ) 263-4333
(Registrant’s telephone number, include area code)
N/A
(Former name or former address, if changed since last report)
——————————————
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |||||
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |||||
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |||||
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) | |||||
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02 Results of Operations and Financial Condition.
On November 10, 2025, Zentalis Pharmaceuticals, Inc. (the “Company”) announced its financial results for the quarter ended September 30, 2025, and commented on certain business updates. The full text of the press release issued in connection with the announcement is furnished as Exhibit 99.1 to this Current Report on Form 8-K (this "Current Report") and is incorporated herein by reference.
Item 7.01 Regulation FD Disclosure.
Beginning November 10, 2025, spokespersons of the Company plan to present the information in the Corporate Presentation furnished as Exhibit 99.2 to this Current Report and incorporated herein by reference at conferences and in meetings with investors and analysts.
The information in Items 2.02 and 7.01 of this Current Report (including Exhibits 99.1 and 99.2 attached hereto) shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Exchange Act or the Securities Act of 1933, as amended, whether made before or after the date hereof, except as expressly set forth by specific reference in such a filing.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits
The following exhibit relating to Item 2.02 shall be deemed to be furnished, and not filed:
Exhibit No. | Description | |||||||
| 99.1 | ||||||||
99.2 | ||||||||
| 104 | Cover Page Interactive Data File (embedded within the inline XBRL document) | |||||||
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| ZENTALIS PHARMACEUTICALS, INC. | ||||||||||||||
| Date: November 10, 2025 | By: | /s/ Julie Eastland | ||||||||||||
| Julie Eastland | ||||||||||||||
| President and Chief Executive Officer | ||||||||||||||

Exhibit 99.1
Zentalis Pharmaceuticals Reports Third Quarter 2025 Financial Results and Operational Progress
DENALI Phase 2 trial evaluating azenosertib in patients with Cyclin E1-positive PROC remains on track with topline data anticipated by year end 2026, with the potential to support an accelerated approval, subject to FDA feedback
$280.7 million cash, cash equivalents and marketable securities supports runway into late 2027
SAN DIEGO, Calif. — November 10, 2025 — Zentalis® Pharmaceuticals, Inc. (Nasdaq: ZNTL), a clinical-stage biopharmaceutical company developing a potentially first-in-class WEE1 inhibitor for patients with ovarian cancer and other tumor types, announced financial results for the third quarter 2025 and highlighted recent operational progress.
“We are pleased with our continued disciplined execution of the DENALI clinical trial this quarter, supporting late-stage development of azenosertib as a potential treatment for Cyclin E1-positive platinum-resistant ovarian cancer, and positioning us for an anticipated topline data readout by year end 2026. Our engagement with trial investigators and presence at medical conferences is very encouraging and continues to support our development strategy," said Julie Eastland, Chief Executive Officer of Zentalis. "With $280.7 million in cash providing runway into late 2027, we maintain a robust financial foundation to deliver on our azenosertib objectives."
Business Updates
•Phase 2 DENALI clinical trial remains on track and has the potential to support an accelerated approval, subject to FDA feedback.
◦Enrollment is ongoing in DENALI Part 2a of the Phase 2 DENALI clinical trial (NCT05128825) of azenosertib in patients with Cyclin E1-positive PROC. DENALI Part 2a is designed to confirm the primary dose-of-interest with a target enrollment of up to approximately 30 patients at each of two dose levels: 400mg QD 5:2 (intermittent daily dosing with a five days on, two days off dosing schedule) and 300mg QD 5:2. DENALI Part 2b is designed to enroll approximately 70 patients at a single dose, the selection of which will be informed by the Part 2a results.
◦The Company expects to disclose topline data from DENALI Part 2 (Part 2a and Part 2b) by year end 2026. We believe that DENALI Part 2, if successful, has the potential to support an accelerated approval, subject to FDA review.
•Poster Presentations at AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics support Cyclin E1 biomarker-driven strategy for azenosertib.
◦Presentations feature data from first-in-human Phase 1 study, including Cyclin E1 biomarker findings, supporting late-stage development of azenosertib.
•TETON Phase 2 trial in uterine serous carcinoma (USC) completed enrollment.
◦Consistent with the Company's previously announced strategic prioritization of azenosertib for the treatment of patients with Cyclin E1-positive PROC, further development in USC will be limited to partnering or the Company's ability to allocate capital to this indication.

◦The Company will continue to support an ongoing investigator-initiated study to explore potential biomarker enrichment strategy in USC.
◦Results from the TETON trial are planned for publication in the first half of 2026.
Third Quarter 2025 Financial Results
•Cash, Cash Equivalents and Marketable Securities Position: As of September 30, 2025, the Company had cash, cash equivalents and marketable securities of $280.7 million. The Company believes that its existing cash, cash equivalents and marketable securities as of September 30, 2025 will be sufficient to fund its operating expenses requirements into late 2027.
•Research and Development Expenses: Research and development (R&D) expenses for the three months ended September 30, 2025 were $23.0 million, compared to $36.8 million for the three months ended September 30, 2024. The decrease of $13.8 million was primarily due to decreases of $7.6 million for personnel expenses, of which $2.7 million was non-cash stock-based compensation. Decreases of $4.2 million for lab services, $1.2 million for clinical expenses, and $0.8 million for supplies, overhead, and other expense also contributed to the overall reduction in research and development expenses.
•General and Administrative Expenses: General and administrative expenses for the three months ended September 30, 2025 were $10.8 million, compared to $14.6 million during the three months ended September 30, 2024. This decrease of $3.8 million was attributable to a decrease of $3.8 million in personnel expense, of which $2.8 million was non-cash stock-based compensation.
•Operating Expenses: Total operating expenses were $33.7 million for the three months ended September 30, 2025, compared to $51.4 million for the three months ended September 30, 2024.
About Azenosertib
Azenosertib is an investigational, novel, selective, and orally bioavailable inhibitor of WEE1 currently being evaluated as a monotherapy and combination clinical studies in ovarian cancer and additional tumor types. WEE1 acts as a master regulator of the G1-S and G2-M cell cycle checkpoints, through negative regulation of both CDK1 and CDK2, to prevent replication of cells with damaged DNA. By inhibiting WEE1, azenosertib enables cell cycle progression, despite high levels of DNA damage, thereby resulting in the accumulation of DNA damage and leading to mitotic catastrophe and cancer cell death.
About DENALI Clinical Trial
DENALI is a multi-part Phase 2 clinical trial studying azenosertib in platinum-resistant ovarian cancer (PROC) patients. Part 1b enrolled patients with PROC regardless of Cyclin E1 protein expression, all treated at 400mg 5:2 (intermittent daily dosing with a five days on, two days off dosing schedule). Interim results from Part 1b were presented at the Society of Gynecologic Oncology (SGO) 2025 Annual Meeting. Part 2 is ongoing and is enrolling PROC patients with Cyclin E1 protein overexpression based on Zentalis’ proprietary immunohistochemistry cutoff. Part 2 includes Part 2a, a dose confirmation portion evaluating two doses, 300mg 5:2 and 400mg 5:2, and Part 2b, a portion designed to complete enrollment at the selected dose. Part 2, in total, is designed for accelerated approval, pending study outcome and discussions with the U.S. Food and Drug Administration.
About Zentalis Pharmaceuticals
Zentalis® Pharmaceuticals, Inc. is a clinical-stage biopharmaceutical company developing azenosertib (ZN-c3), an investigational, potentially first-in-class and best-in-class WEE1 inhibitor for patients with Cyclin E1-positive platinum-resistant ovarian cancer (PROC). Azenosertib is being evaluated as a

monotherapy and in combination across multiple tumor types in clinical trials and has broad franchise potential. In clinical trials, azenosertib has been well tolerated and has demonstrated anti-tumor activity as a single agent across multiple tumor types. The Company is also leveraging its extensive experience and capabilities to translate its science to advance research on additional areas of opportunity for azenosertib outside PROC. Zentalis has operations in San Diego.
For more information, please visit www.zentalis.com. Follow Zentalis on LinkedIn at www.linkedin.com/company/zentalis-pharmaceuticals
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, as amended. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including, but not limited to, statements regarding the potential of azenosertib; our anticipated milestones and the timing thereof, including the anticipated timing of clinical data disclosures from DENALI Part 2 and TETON; the potential to advance research on additional areas of opportunity for azenosertib outside PROC, including any further development in USC; our anticipated cash runway; the potential for azenosertib to be first-in-class; the broad franchise potential of azenosertib; the planned design of our clinical trials, including DENALI Part 2; maintaining momentum and remaining on track relating to the execution of DENALI; and our planned clinical development strategy and regulatory strategy for azenosertib and the timing thereof, including the potential for DENALI Part 2 to support an accelerated approval. The terms “advance,” “anticipated,” “believe,” “continue,” “design,” “expect,” “opportunity,” “on track,” “plan,” “position,” “potential,” “runway,” “target,” and “will” and similar references are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the following: our limited operating history, which may make it difficult to evaluate our current business and predict our future success and viability; we have and expect to continue to incur significant losses; our need for additional funding, which may not be available; our substantial dependence on the success of azenosertib; our plans, including the costs thereof, of development of companion diagnostics; the outcome of preclinical testing and early trials may not be predictive of the success of later clinical trials; failure to identify additional product candidates and develop or commercialize marketable products; potential unforeseen events during clinical trials could cause delays or other adverse consequences; risks relating to the regulatory approval process or ongoing regulatory obligations; failure to obtain U.S. or international marketing approval; our product candidates may cause serious adverse side effects; inability to maintain our collaborations, or the failure of these collaborations; our reliance on third parties; effects of significant competition; the possibility of system failures or security breaches; risks relating to intellectual property; our ability to attract, retain and motivate qualified personnel, and risks relating to management transitions; significant costs as a result of operating as a public company; and the other important factors discussed under the caption “Risk Factors” in our most recently filed periodic report on Form 10-K or 10-Q and subsequent filings with the U.S. Securities and Exchange Commission (SEC) and our other filings with the SEC. Any such forward-looking statements represent management’s estimates as of the date of this press release. While we may elect to update such forward-looking statements at some point in the future, we disclaim any obligation to do so, even if subsequent events cause our views to change.
ZENTALIS® and its associated logo are trademarks of Zentalis and/or its affiliates. All website addresses and other links in this press release are for information only and are not intended to be an active link or to incorporate any website or other information into this press release.

| Zentalis Pharmaceuticals, Inc. | |||||||||||||||||||||||
| Condensed Consolidated Statements of Operations | |||||||||||||||||||||||
| (Unaudited) | |||||||||||||||||||||||
| (In thousands, except per share amounts) | |||||||||||||||||||||||
| Three Months Ended September 30, | Nine Months Ended September 30, | ||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||
| License Revenue | $ | — | $ | — | $ | — | $ | 40,560 | |||||||||||||||
| Operating Expenses | |||||||||||||||||||||||
| Research and development | 22,950 | 36,824 | 77,807 | 134,795 | |||||||||||||||||||
| General and administrative | 10,784 | 14,608 | 29,812 | 47,110 | |||||||||||||||||||
| Restructuring | — | — | 7,796 | — | |||||||||||||||||||
| Total operating expenses | 33,734 | 51,432 | 115,415 | 181,905 | |||||||||||||||||||
| Loss from Operations | (33,734) | (51,432) | (115,415) | (141,345) | |||||||||||||||||||
| Other Income (Expense) | |||||||||||||||||||||||
| Investment and other income, net | 7,043 | 11,247 | 13,571 | 23,332 | |||||||||||||||||||
| Net loss before income taxes | (26,691) | (40,185) | (101,844) | (118,013) | |||||||||||||||||||
| Income tax expense (benefit) | — | (27) | — | 382 | |||||||||||||||||||
| Net loss | (26,691) | (40,158) | (101,844) | (118,395) | |||||||||||||||||||
| Net loss attributable to noncontrolling interests | — | — | — | (28) | |||||||||||||||||||
| Net loss attributable to Zentalis | $ | (26,691) | $ | (40,158) | $ | (101,844) | $ | (118,367) | |||||||||||||||
| Net loss per share outstanding, basic and diluted | $ | (0.37) | $ | (0.56) | $ | (1.42) | $ | (1.67) | |||||||||||||||
| Common shares used in computing net loss per share, basic and diluted | 72,139 | 71,111 | 71,938 | 71,017 | |||||||||||||||||||

| Zentalis Pharmaceuticals, Inc. | ||||||||||||||
| Selected Condensed Consolidated Balance Sheet Data | ||||||||||||||
| (Unaudited) | ||||||||||||||
| (In thousands) | ||||||||||||||
| As of September 30, | As of December 31, | |||||||||||||
| 2025 | 2024 | |||||||||||||
| Cash, cash equivalents and marketable securities | $ | 280,697 | $ | 371,084 | ||||||||||
Working capital (1) | 251,134 | 333,341 | ||||||||||||
| Total assets | 327,250 | 430,337 | ||||||||||||
| Total liabilities | 74,376 | 93,151 | ||||||||||||
| Total Zentalis equity | $ | 252,874 | $ | 337,186 | ||||||||||
(1) The Company defines working capital as current assets less current liabilities. | ||||||||||||||
Contact:
Aron Feingold
VP, Investor Relations & Corporate Communications
ir@zentalis.com

Corporate Presentation November 2025 Nasdaq: ZNTL

2 Zentalis Pharmaceuticals, Inc. (“we,” “us,” “our,” “Zentalis” or the “Company”) cautions that this presentation (including oral commentary that accompanies this presentation) contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995. All statements contained in this presentation that do not relate to matters of historical fact should be considered forward-looking statements, including without limitation statements regarding the potential for azenosertib (ZN-c3) to be first-in-class and best-in-class; the potential regulatory strategy, approach and pathway for azenosertib, including the potential for FDA accelerated approval of azenosertib in Cyclin E1-positive PROC and the potential for studies to be registrational or intended for registration; our development strategy and approach for azenosertib, including our strategy to focus on bringing azenosertib to patients with PROC who are Cyclin E1-positive and the potential for azenosertib to be a new treatment option for ovarian cancer patients; our planned strategy, vision and path forward; the market opportunity for azenosertib, including the opportunity in biomarker selected (Cyclin E1-positive) PROC patients and the potential size of the patient population; existing data being supportive of the go-forward azenosertib development strategy; the potential for the opportunity for azenosertib to be broad/expansive; the potential opportunities for azenosertib as a monotherapy and in combination in other indications and in other tumor types, including in earlier lines of ovarian cancer, breast, endometrial and bladder; the potential for Cyclin E1 to serve as a predictive biomarker for response to azenosertib; the opportunity to improve outcomes in the next stage of development, including via close monitoring; our projected cash runway; planned clinical trials for our product candidates, including the Phase 3 confirmatory trial of azenosertib in Cyclin E1-positive PROC; the potential of azenosertib to address a significant unmet need in patients with PROC who are Cyclin E1-positive; the potential benefits of azenosertib, including compared to available therapies and therapies in development (not head-to-head comparisons); the potential unmet need in a particular indication and/or patient population; the timing and content of our anticipated milestones, including enrollment of patients, disclosure of clinical data, and initiation of the Phase 3 confirmatory trial for azenosertib in Cyclin E1-positive PROC; as well as statements that include the words such as “approximately,” “anticipate,” “beyond,” “estimate,” “expect,” “forward,” “intent,” “milestone,” “ongoing,” “opportunity,” “path,” “plan,” “potential,” “predictive,” “projected,” “strategy,” "support," “vision,” “will” and similar statements of a future or forward-looking nature. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the following: our limited operating history, which may make it difficult to evaluate our current business and predict our future success and viability; we have and expect to continue to incur significant losses; our need for additional funding, which may not be available; our plans, including the costs thereof, of development of a companion diagnostic; the outcome of preclinical testing and early trials may not be predictive of the success of later clinical trials; failure to identify additional product candidates and develop or commercialize marketable products; potential unforeseen events during clinical trials could cause delays or other adverse consequences; risks relating to the regulatory approval process or ongoing regulatory obligations; failure to obtain U.S. or international marketing approval; azenosertib and any future product candidates may cause serious adverse side effects; inability to maintain our collaborations, or the failure of these collaborations; our reliance on third parties; effects of significant competition; the possibility of system failures or security breaches; risks relating to intellectual property; our ability to attract, retain and motivate qualified personnel, and risks relating to management transitions; and significant costs as a result of operating as a public company. Other risks and uncertainties include those identified under the caption “Risk Factors” in our most recently filed periodic reports on Forms 10-K and 10-Q and subsequent filings with the U.S. Securities and Exchange Commission in the future could cause such forward-looking statements represent management’s estimates as of the date of this presentation. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. While we may elect to update these forward-looking statements at some point in the future, we assume no obligation to update or revise any forward-looking statements except to the extent required by applicable actual results to differ materially from those indicated by the forward-looking statements made in this presentation. Any law. Although we believe the expectations reflected in such forward-looking statements are reasonable, we can give no assurance that such expectations will prove to be correct. Accordingly, readers are cautioned not to place undue reliance on these forward-looking statements. No representations or warranties (expressed or implied) are made about the accuracy of any such forward-looking statements. This presentation also contains estimates and other statistical data made by independent parties and by us relating to market size and other data about our industry. These data involve a number of assumptions and limitations, and you are cautioned not to give undue weight to such data and estimates. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. Neither we nor our affiliates, advisors or representatives makes any representation as to the accuracy or completeness of that data or undertake to update such data after the date of this presentation. Statements such as “not head-to-head,” “direct cross-study comparison not intended” and similar references indicate that no head-to-head clinical trial has been conducted evaluating azenosertib against the indicated therapies. Notable differences exist between the Company’s trial designs, conditions under study and subject characteristics as compared to the evaluated third party results and caution should be exercised when comparing data across these studies. ZENTALIS® and its associated logos are trademarks of Zentalis and/or its affiliates. All other trademarks, trade names and service marks appearing in this presentation are the property of their respective owners. All website addresses given in this presentation are for information only and are not intended to be an active link or to incorporate any website information into this document. Azenosertib is an investigational drug and have not yet been approved by the U.S. Food and Drug Administration or any other regulatory authority. Forward Looking Statements and Disclaimer

3 Extensive Data Support Azenosertib as Potential Best-in-Class, Orally Available, Non-Chemo Therapy for Patients with Cyclin E1-Positive Platinum-Resistant Ovarian Cancer (PROC) >30% ORR, ~6 mos mDOR‡ ‡ As of Jan. 13, 2025 data cutoff in DENALI Part 1b, mDOR subject to change; † Not a head-to-head comparison; *See integrated monotherapy safety data on slide 12 1. Kang EY, et al. Cancer. 2023;129:697-713 2. Eskander, R., et al. Overcoming the Challenges in Drug Development, Front Oncol. 2023 Oct 17; 13:1258228 Abbreviations: ORR = objective response rate; mDOR = median duration of response; 5:2 schedule = 5 days once-daily administration of azenosertib, followed by 2 days without azenosertib in Cyclin E1-positive PROC patients at monotherapy dose of 400mg QD 5:2 350+ patients treated at active doses in monotherapy 200+ PROC patients treated at monotherapy 300mg and 400mg QD 5:2 Manageable safety profile* Safety and tolerability at 300 and 400 mg QD 5:2 broadly comparable Cyclin E1 protein overexpression is a biomarker of poor prognosis and low benefit from standard-of- care (SOC) single-agent chemotherapy in PROC1 4-13% ORR for SOC mono chemo in PROC reported in literature2†

4 Projected Cash Runway into Late 2027, Beyond Anticipated Registration- Intent Phase 2 Topline Data o 2026: Initiate Phase 3 confirmatory trial following FDA feedback o YE 2026: Topline data from registration-intent† DENALI Part 2 o Potential for FDA accelerated approval in Cyclin E1- Positive PROC patients† Anticipated Upcoming Milestones * Cyclin E1 IHC+ based on Zentalis proprietary IHC cutoff and Cyclin E1 IHC assay developed from the existing clinical data † Subject to supportive data and FDA feedback and approval Abbreviations: 5:2 schedule = 5 days once-daily administration of azenosertib, followed by 2 days without azenosertib $280.7M cash, cash equivalents and marketable securities as of September 30, 2025 Interim clinical data demonstrated clinically meaningful results and manageable safety profile across multiple azenosertib monotherapy studies, with 200+ PROC patients treated at 300mg and 400mg QD 5:2 Established Cyclin E1 protein overexpression, as determined by proprietary IHC cutoff*, as predictive biomarker for azenosertib in PROC Registration-intent† Part 2 of the Phase 2 DENALI clinical trial is underway and enrolling patients Strategic Focus and Resource Prioritization on Cyclin E1-Positive PROC
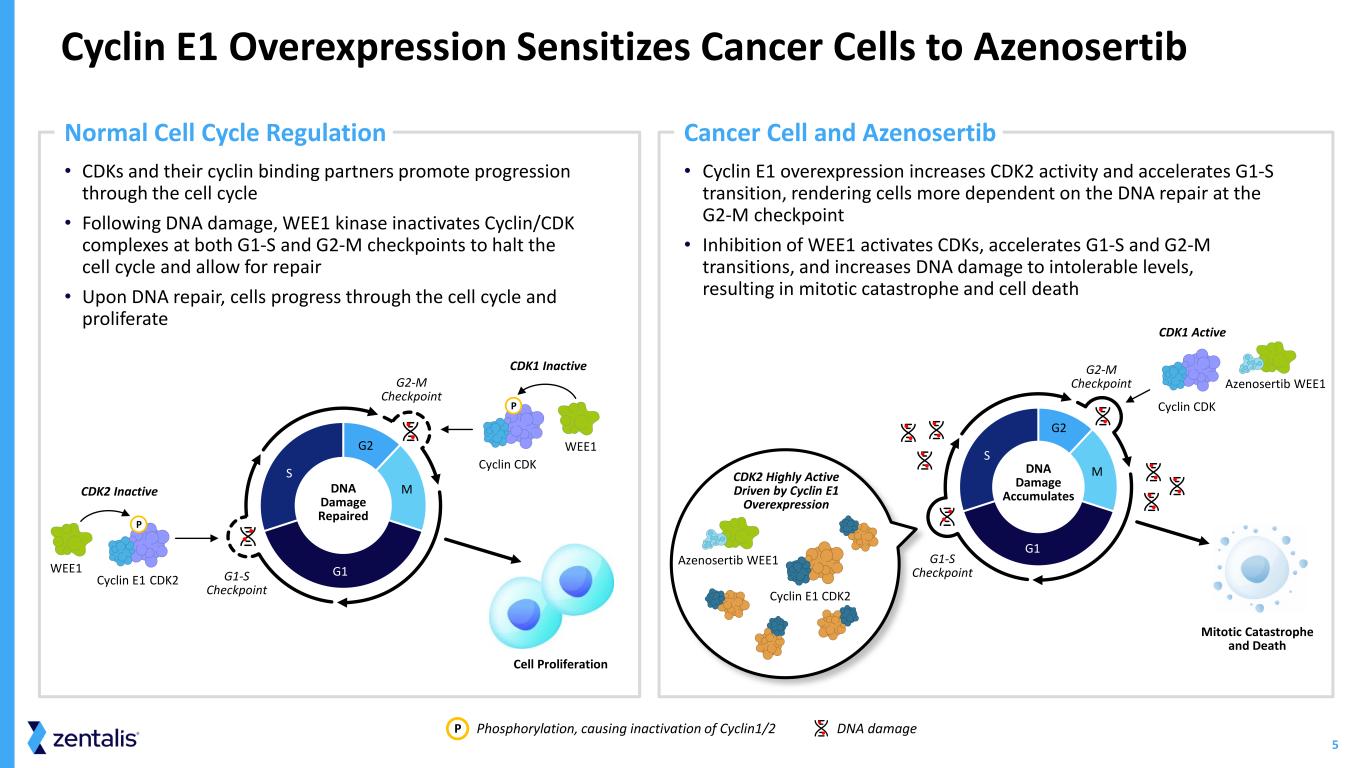
5 • Cyclin E1 overexpression increases CDK2 activity and accelerates G1-S transition, rendering cells more dependent on the DNA repair at the G2-M checkpoint • Inhibition of WEE1 activates CDKs, accelerates G1-S and G2-M transitions, and increases DNA damage to intolerable levels, resulting in mitotic catastrophe and cell death • CDKs and their cyclin binding partners promote progression through the cell cycle • Following DNA damage, WEE1 kinase inactivates Cyclin/CDK complexes at both G1-S and G2-M checkpoints to halt the cell cycle and allow for repair • Upon DNA repair, cells progress through the cell cycle and proliferate Cyclin E1 Overexpression Sensitizes Cancer Cells to Azenosertib Phosphorylation, causing inactivation of Cyclin1/2 DNA damageP G1 S G2 MDNA Damage Repaired CDK2 Inactive CDK2Cyclin E1 WEE1 P CDK1 Inactive WEE1 Cell Proliferation P Normal Cell Cycle Regulation Cancer Cell and Azenosertib Mitotic Catastrophe and Death G2-M Checkpoint DNA Damage Accumulates CDK2 Highly Active Driven by Cyclin E1 Overexpression Cyclin CDK Azenosertib WEE1 Azenosertib WEE1 CDK1 Active G1-S Checkpoint Cyclin E1 CDK2 G1 S G2 MCyclin CDK G2-M Checkpoint G1-S Checkpoint
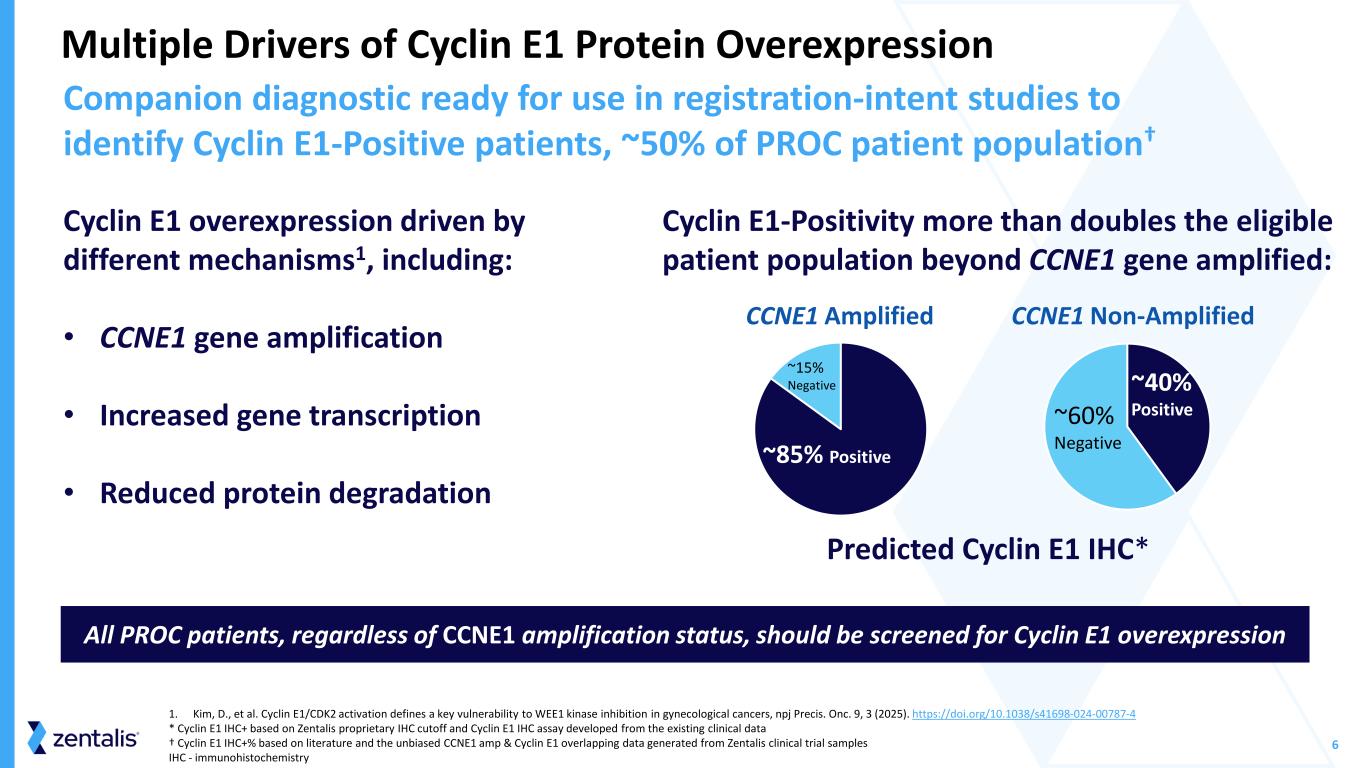
6 Multiple Drivers of Cyclin E1 Protein Overexpression Companion diagnostic ready for use in registration-intent studies to identify Cyclin E1-Positive patients, ~50% of PROC patient population† 1. Kim, D., et al. Cyclin E1/CDK2 activation defines a key vulnerability to WEE1 kinase inhibition in gynecological cancers, npj Precis. Onc. 9, 3 (2025). https://doi.org/10.1038/s41698-024-00787-4 * Cyclin E1 IHC+ based on Zentalis proprietary IHC cutoff and Cyclin E1 IHC assay developed from the existing clinical data † Cyclin E1 IHC+% based on literature and the unbiased CCNE1 amp & Cyclin E1 overlapping data generated from Zentalis clinical trial samples IHC - immunohistochemistry All PROC patients, regardless of CCNE1 amplification status, should be screened for Cyclin E1 overexpression Cyclin E1 overexpression driven by different mechanisms1, including: • CCNE1 gene amplification • Increased gene transcription • Reduced protein degradation Cyclin E1-Positivity more than doubles the eligible patient population beyond CCNE1 gene amplified: ~15% Negative ~85% Positive CCNE1 Non-Amplified Predicted Cyclin E1 IHC * CCNE1 Amplified ~40% Positive~60% Negative
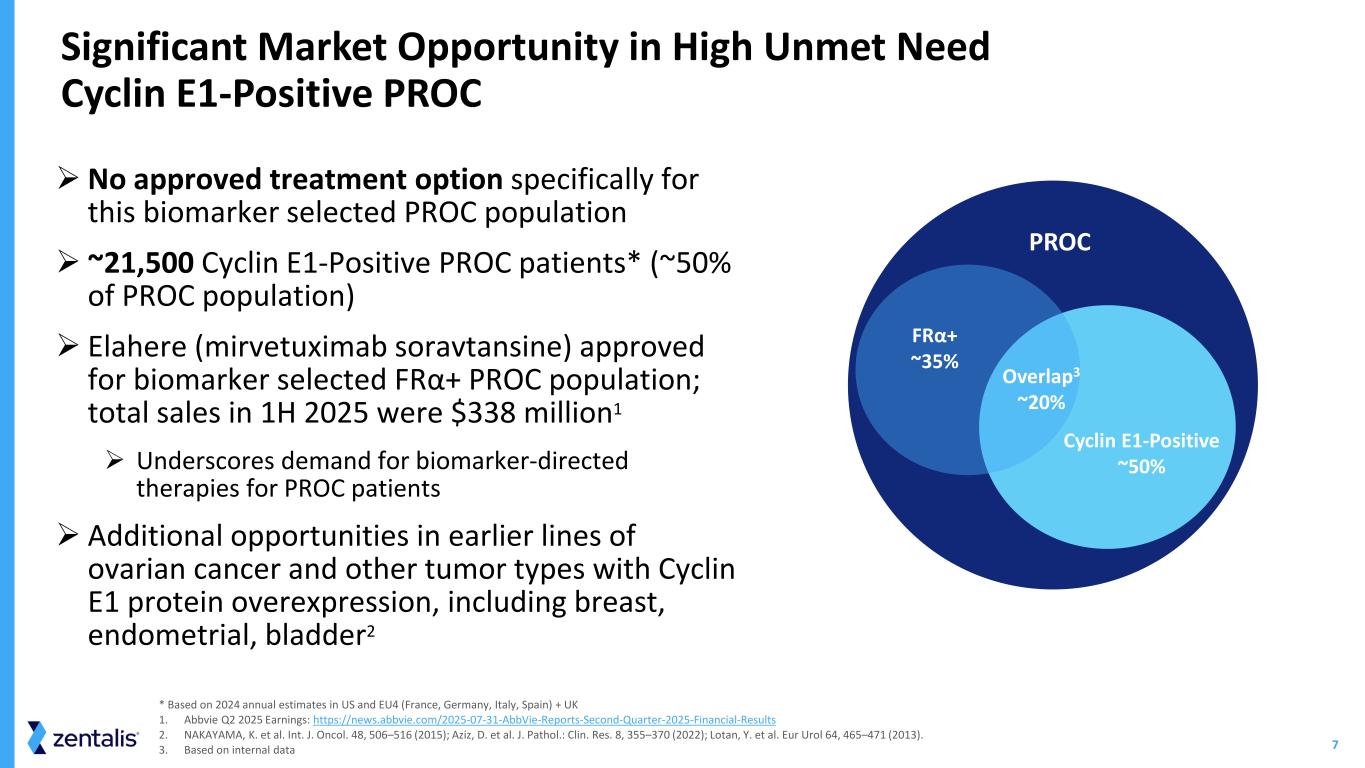
7 Significant Market Opportunity in High Unmet Need Cyclin E1-Positive PROC * Based on 2024 annual estimates in US and EU4 (France, Germany, Italy, Spain) + UK 1. Abbvie Q2 2025 Earnings: https://news.abbvie.com/2025-07-31-AbbVie-Reports-Second-Quarter-2025-Financial-Results 2. NAKAYAMA, K. et al. Int. J. Oncol. 48, 506–516 (2015); Aziz, D. et al. J. Pathol.: Clin. Res. 8, 355–370 (2022); Lotan, Y. et al. Eur Urol 64, 465–471 (2013). 3. Based on internal data No approved treatment option specifically for this biomarker selected PROC population ~21,500 Cyclin E1-Positive PROC patients* (~50% of PROC population) Elahere (mirvetuximab soravtansine) approved for biomarker selected FRα+ PROC population; total sales in 1H 2025 were $338 million1 Underscores demand for biomarker-directed therapies for PROC patients Additional opportunities in earlier lines of ovarian cancer and other tumor types with Cyclin E1 protein overexpression, including breast, endometrial, bladder2 3 PROC FRα+ ~35% Overlap3 ~20% Cyclin E1-Positive ~50%
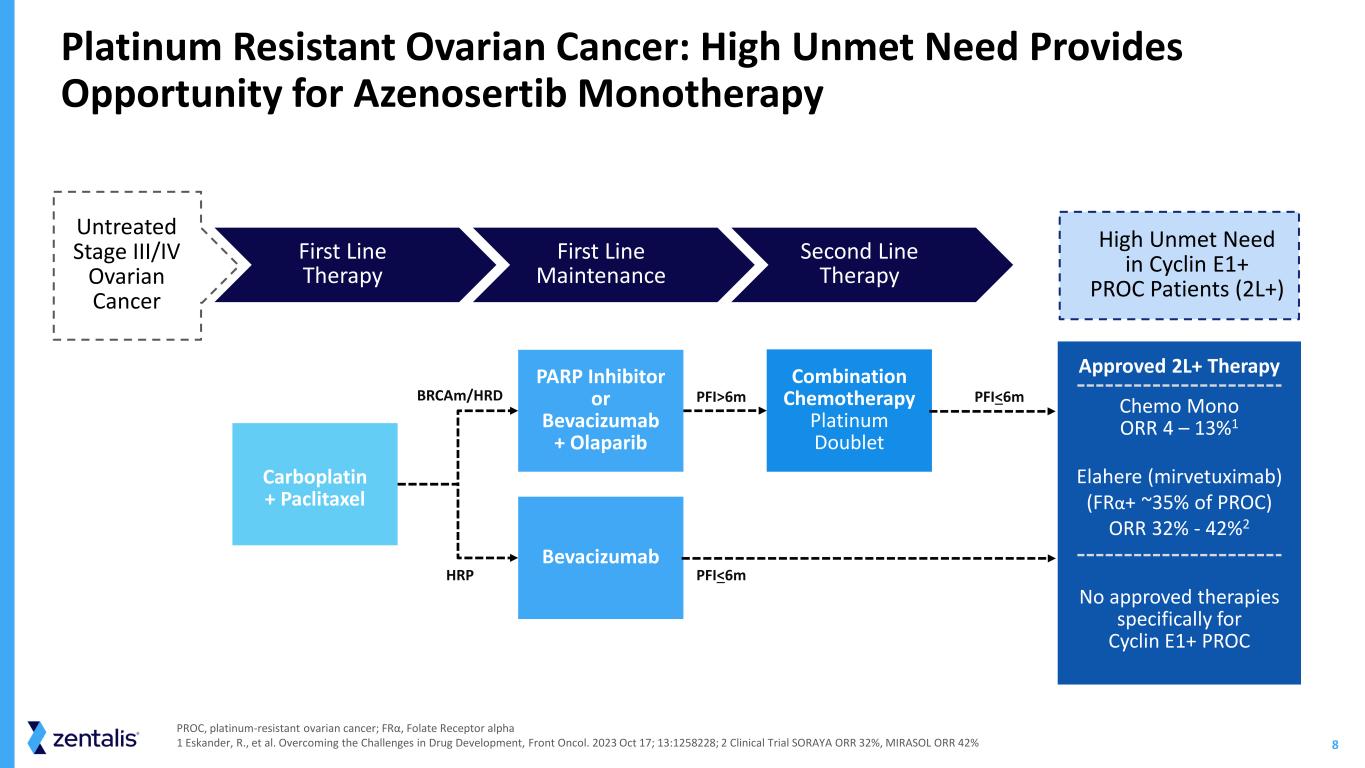
8 PARP Inhibitor or Bevacizumab + Olaparib Bevacizumab Combination Chemotherapy Platinum Doublet HRP PFI>6m PFI<6m PFI<6m Carboplatin + Paclitaxel High Unmet Need in Cyclin E1+ PROC Patients (2L+) Approved 2L+ Therapy Chemo Mono ORR 4 – 13%1 Elahere (mirvetuximab) (FRα+ ~35% of PROC) ORR 32% - 42%2 No approved therapies specifically for Cyclin E1+ PROC Platinum Resistant Ovarian Cancer: High Unmet Need Provides Opportunity for Azenosertib Monotherapy First Line Maintenance Second Line Therapy First Line Therapy BRCAm/HRD Untreated Stage III/IV Ovarian Cancer PROC, platinum-resistant ovarian cancer; FRα, Folate Receptor alpha 1 Eskander, R., et al. Overcoming the Challenges in Drug Development, Front Oncol. 2023 Oct 17; 13:1258228; 2 Clinical Trial SORAYA ORR 32%, MIRASOL ORR 42%
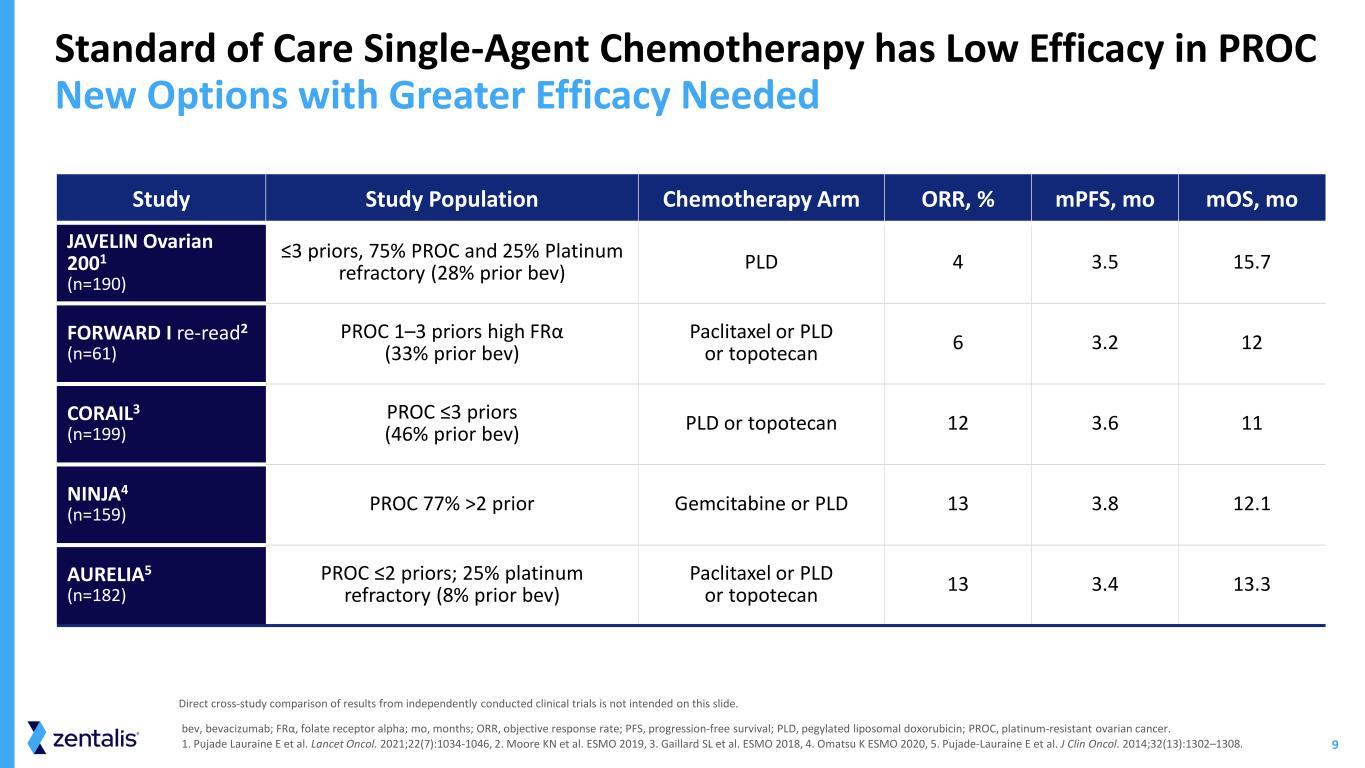
9 Standard of Care Single-Agent Chemotherapy has Low Efficacy in PROC New Options with Greater Efficacy Needed bev, bevacizumab; FRα, folate receptor alpha; mo, months; ORR, objective response rate; PFS, progression-free survival; PLD, pegylated liposomal doxorubicin; PROC, platinum-resistant ovarian cancer. 1. Pujade Lauraine E et al. Lancet Oncol. 2021;22(7):1034-1046, 2. Moore KN et al. ESMO 2019, 3. Gaillard SL et al. ESMO 2018, 4. Omatsu K ESMO 2020, 5. Pujade-Lauraine E et al. J Clin Oncol. 2014;32(13):1302–1308. Study Study Population Chemotherapy Arm ORR, % mPFS, mo mOS, mo JAVELIN Ovarian 2001 (n=190) ≤3 priors, 75% PROC and 25% Platinum refractory (28% prior bev) PLD 4 3.5 15.7 FORWARD I re-read2 (n=61) PROC 1–3 priors high FRα (33% prior bev) Paclitaxel or PLD or topotecan 6 3.2 12 CORAIL3 (n=199) PROC ≤3 priors (46% prior bev) PLD or topotecan 12 3.6 11 NINJA4 (n=159) PROC 77% >2 prior Gemcitabine or PLD 13 3.8 12.1 AURELIA5 (n=182) PROC ≤2 priors; 25% platinum refractory (8% prior bev) Paclitaxel or PLD or topotecan 13 3.4 13.3 Direct cross-study comparison of results from independently conducted clinical trials is not intended on this slide.

Integrated Data and Potential Path to Registration in PROC
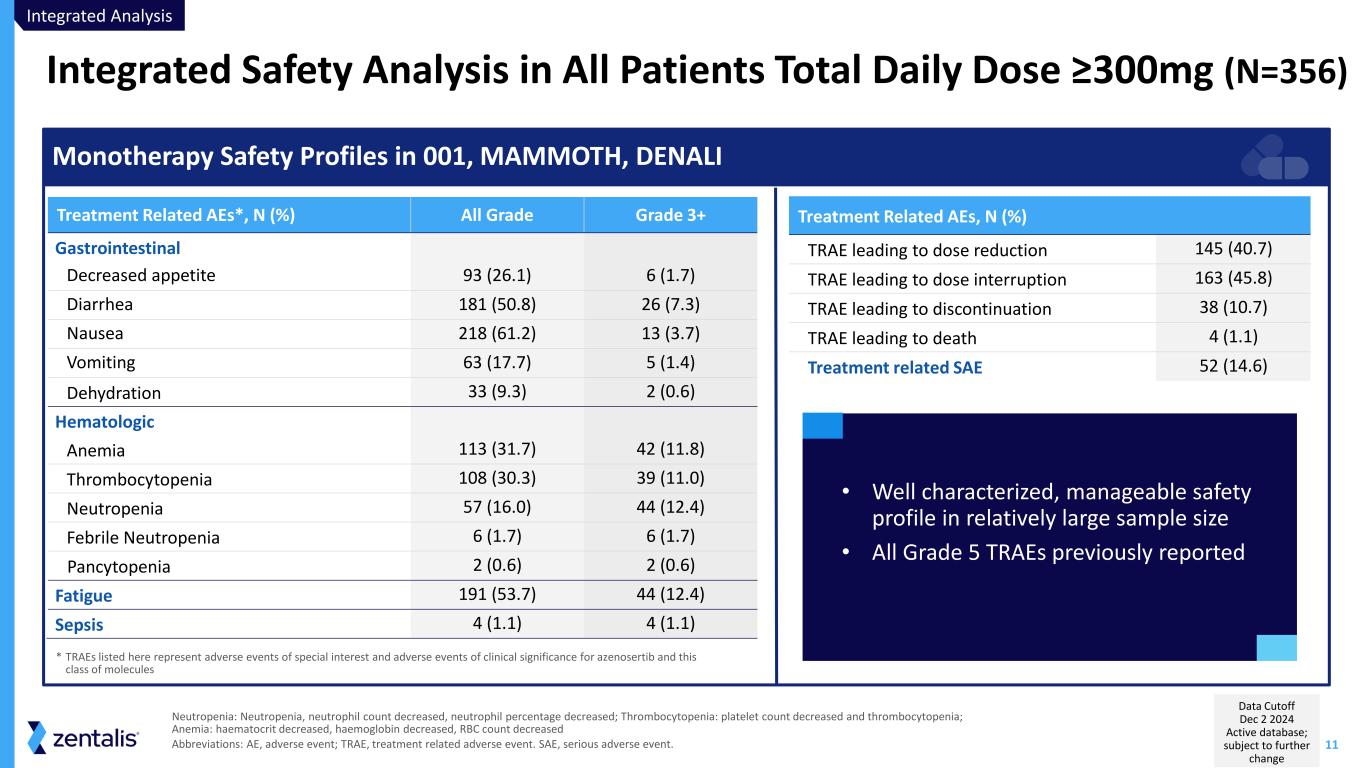
11 Integrated Safety Analysis in All Patients Total Daily Dose ≥300mg (N=356) Neutropenia: Neutropenia, neutrophil count decreased, neutrophil percentage decreased; Thrombocytopenia: platelet count decreased and thrombocytopenia; Anemia: haematocrit decreased, haemoglobin decreased, RBC count decreased Abbreviations: AE, adverse event; TRAE, treatment related adverse event. SAE, serious adverse event. Monotherapy Safety Profiles in 001, MAMMOTH, DENALI Treatment Related AEs, N (%) TRAE leading to dose reduction 145 (40.7) TRAE leading to dose interruption 163 (45.8) TRAE leading to discontinuation 38 (10.7) TRAE leading to death 4 (1.1) Treatment related SAE 52 (14.6) Treatment Related AEs*, N (%) All Grade Grade 3+ Gastrointestinal Decreased appetite 93 (26.1) 6 (1.7) Diarrhea 181 (50.8) 26 (7.3) Nausea 218 (61.2) 13 (3.7) Vomiting 63 (17.7) 5 (1.4) Dehydration 33 (9.3) 2 (0.6) Hematologic Anemia 113 (31.7) 42 (11.8) Thrombocytopenia 108 (30.3) 39 (11.0) Neutropenia 57 (16.0) 44 (12.4) Febrile Neutropenia 6 (1.7) 6 (1.7) Pancytopenia 2 (0.6) 2 (0.6) Fatigue 191 (53.7) 44 (12.4) Sepsis 4 (1.1) 4 (1.1) • Well characterized, manageable safety profile in relatively large sample size • All Grade 5 TRAEs previously reported * TRAEs listed here represent adverse events of special interest and adverse events of clinical significance for azenosertib and this class of molecules Data Cutoff Dec 2 2024 Active database; subject to further change Integrated Analysis
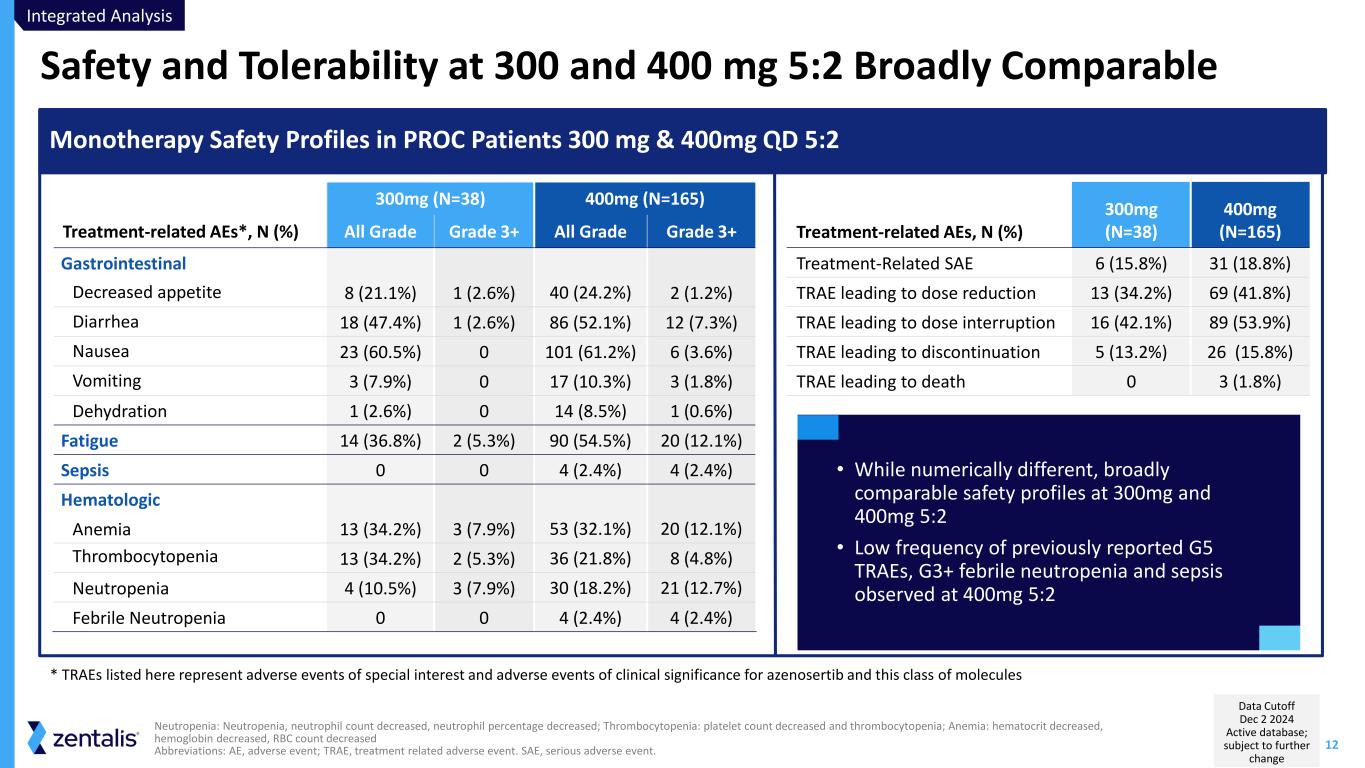
12 Safety and Tolerability at 300 and 400 mg 5:2 Broadly Comparable Neutropenia: Neutropenia, neutrophil count decreased, neutrophil percentage decreased; Thrombocytopenia: platelet count decreased and thrombocytopenia; Anemia: hematocrit decreased, hemoglobin decreased, RBC count decreased Abbreviations: AE, adverse event; TRAE, treatment related adverse event. SAE, serious adverse event. 300mg (N=38) 400mg (N=165) Treatment-related AEs*, N (%) All Grade Grade 3+ All Grade Grade 3+ Gastrointestinal Decreased appetite 8 (21.1%) 1 (2.6%) 40 (24.2%) 2 (1.2%) Diarrhea 18 (47.4%) 1 (2.6%) 86 (52.1%) 12 (7.3%) Nausea 23 (60.5%) 0 101 (61.2%) 6 (3.6%) Vomiting 3 (7.9%) 0 17 (10.3%) 3 (1.8%) Dehydration 1 (2.6%) 0 14 (8.5%) 1 (0.6%) Fatigue 14 (36.8%) 2 (5.3%) 90 (54.5%) 20 (12.1%) Sepsis 0 0 4 (2.4%) 4 (2.4%) Hematologic Anemia 13 (34.2%) 3 (7.9%) 53 (32.1%) 20 (12.1%) Thrombocytopenia 13 (34.2%) 2 (5.3%) 36 (21.8%) 8 (4.8%) Neutropenia 4 (10.5%) 3 (7.9%) 30 (18.2%) 21 (12.7%) Febrile Neutropenia 0 0 4 (2.4%) 4 (2.4%) Monotherapy Safety Profiles in PROC Patients 300 mg & 400mg QD 5:2 Treatment-related AEs, N (%) 300mg (N=38) 400mg (N=165) Treatment-Related SAE 6 (15.8%) 31 (18.8%) TRAE leading to dose reduction 13 (34.2%) 69 (41.8%) TRAE leading to dose interruption 16 (42.1%) 89 (53.9%) TRAE leading to discontinuation 5 (13.2%) 26 (15.8%) TRAE leading to death 0 3 (1.8%) * TRAEs listed here represent adverse events of special interest and adverse events of clinical significance for azenosertib and this class of molecules • While numerically different, broadly comparable safety profiles at 300mg and 400mg 5:2 • Low frequency of previously reported G5 TRAEs, G3+ febrile neutropenia and sepsis observed at 400mg 5:2 Integrated Analysis Data Cutoff Dec 2 2024 Active database; subject to further change
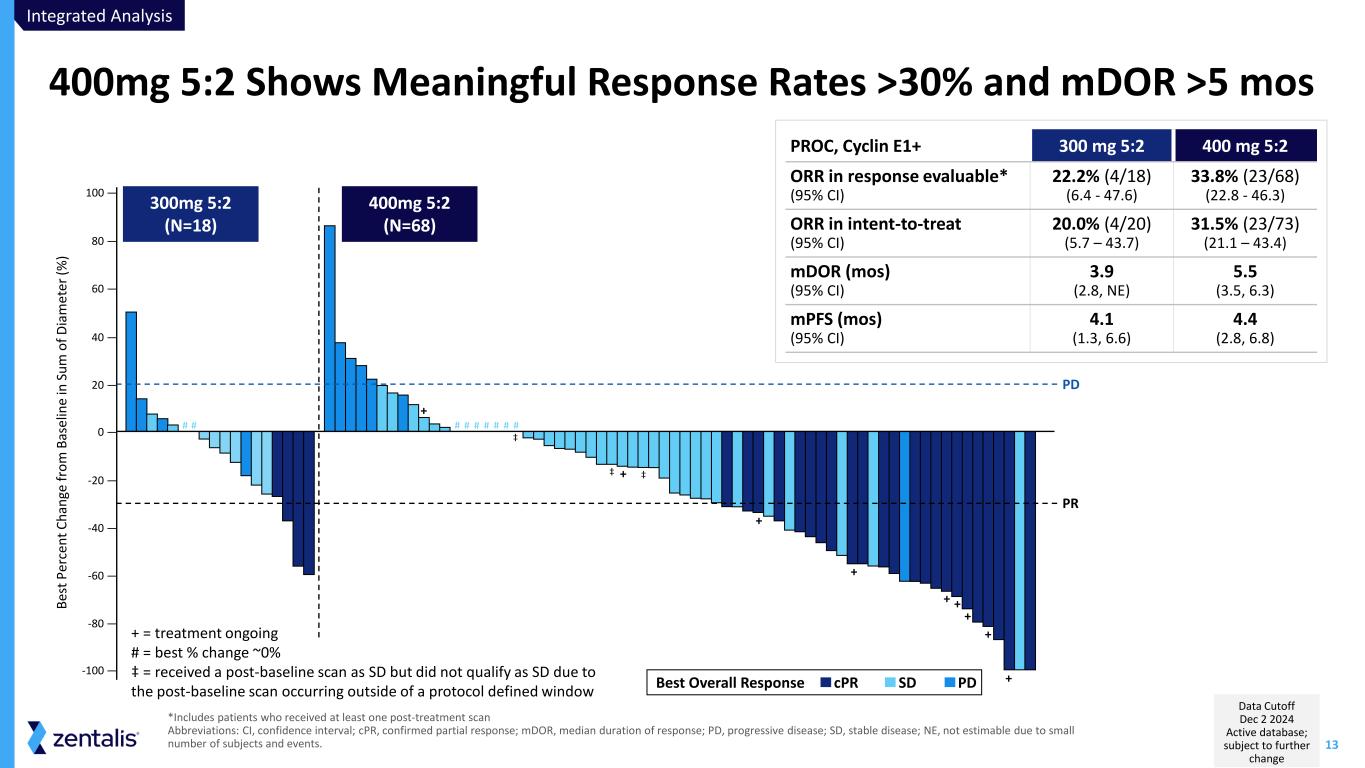
13 *Includes patients who received at least one post-treatment scan Abbreviations: CI, confidence interval; cPR, confirmed partial response; mDOR, median duration of response; PD, progressive disease; SD, stable disease; NE, not estimable due to small number of subjects and events. 400mg 5:2 Shows Meaningful Response Rates >30% and mDOR >5 mos + + + + + + + + +-100 — -80 — -60 — -40 — -20 — 0 — 20 — 40 — 60 — 80 — 100 — Be st P er ce nt C ha ng e fr om B as el in e in S um o f D ia m et er (% ) PR PD 300mg 5:2 (N=18) 400mg 5:2 (N=68) PDSDcPRBest Overall Response # # # # # # # # # ‡ ‡ ‡ + = treatment ongoing # = best % change ~0% ‡ = received a post-baseline scan as SD but did not qualify as SD due to the post-baseline scan occurring outside of a protocol defined window Integrated Analysis Data Cutoff Dec 2 2024 Active database; subject to further change PROC, Cyclin E1+ 300 mg 5:2 400 mg 5:2 ORR in response evaluable* (95% CI) 22.2% (4/18) (6.4 - 47.6) 33.8% (23/68) (22.8 - 46.3) ORR in intent-to-treat (95% CI) 20.0% (4/20) (5.7 – 43.7) 31.5% (23/73) (21.1 – 43.4) mDOR (mos) (95% CI) 3.9 (2.8, NE) 5.5 (3.5, 6.3) mPFS (mos) (95% CI) 4.1 (1.3, 6.6) 4.4 (2.8, 6.8)
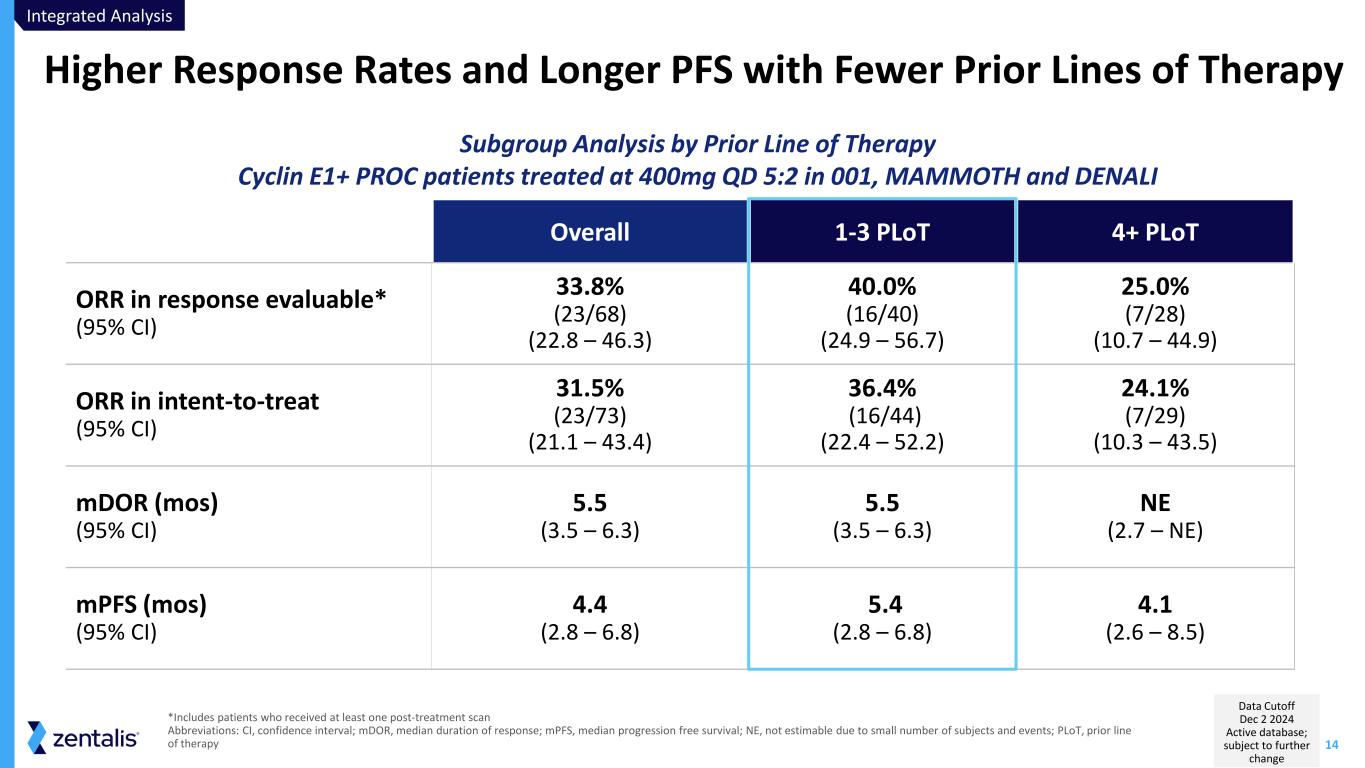
14 Higher Response Rates and Longer PFS with Fewer Prior Lines of Therapy Integrated Analysis Data Cutoff Dec 2 2024 Active database; subject to further change Overall 1-3 PLoT 4+ PLoT ORR in response evaluable* (95% CI) 33.8% (23/68) (22.8 – 46.3) 40.0% (16/40) (24.9 – 56.7) 25.0% (7/28) (10.7 – 44.9) ORR in intent-to-treat (95% CI) 31.5% (23/73) (21.1 – 43.4) 36.4% (16/44) (22.4 – 52.2) 24.1% (7/29) (10.3 – 43.5) mDOR (mos) (95% CI) 5.5 (3.5 – 6.3) 5.5 (3.5 – 6.3) NE (2.7 – NE) mPFS (mos) (95% CI) 4.4 (2.8 – 6.8) 5.4 (2.8 – 6.8) 4.1 (2.6 – 8.5) *Includes patients who received at least one post-treatment scan Abbreviations: CI, confidence interval; mDOR, median duration of response; mPFS, median progression free survival; NE, not estimable due to small number of subjects and events; PLoT, prior line of therapy Subgroup Analysis by Prior Line of Therapy Cyclin E1+ PROC patients treated at 400mg QD 5:2 in 001, MAMMOTH and DENALI

15 Randomized Study vs. Standard of Care Same Key Eligibility as DENALI Part 2 Plan to Conduct Concurrently with DENALI Part 2b Detail Design to be Discussed with FDA Abbreviations: 5:2 schedule = 5 days once-daily administration of azenosertib, followed by 2 days without azenosertib; IHC= immunohistochemistry; MIRV, mirvetuximab soravtansine; PROC=platinum resistant ovarian cancer. DENALI PART 2 FOR POTENTIAL ACCELERATED APPROVAL (N= ~100 at selected dose) PHASE 3 CONFIRMATORY STUDY (Pending FDA feedback) Platinum-resistant ovarian cancer 1-3 prior lines of therapy Prior MIRV if high FRα Cyclin E1+ by proprietary IHC cutoff criteria Key Eligibility Registration-Intent Studies for Azenosertib Monotherapy in PROC, Cyclin E1+ Population Part 2a (Alignment with FDA) Part 2b (Pending FDA feedback) Continuous Enrollment Across Part 2a and 2b with Dose Confirmation at Interim Analysis PROC Pts Cyclin E1+ Tumors 400mg 5:2 (N=30) 300mg 5:2 (N=30) 1:1 Randomization Dose TBD 5:2 (N=~70) Endpoints PFS Safety and Tolerability ORR, DOR Pr es cr ee ni ng / Ti ss ue C on se nt Se am le ss E nr ol lm en t Dose Confirmation

DENALI Part 1b (ZN-c3-005) NCT05198804 Updated data at SGO 2025, Data cutoff January 13, 2025
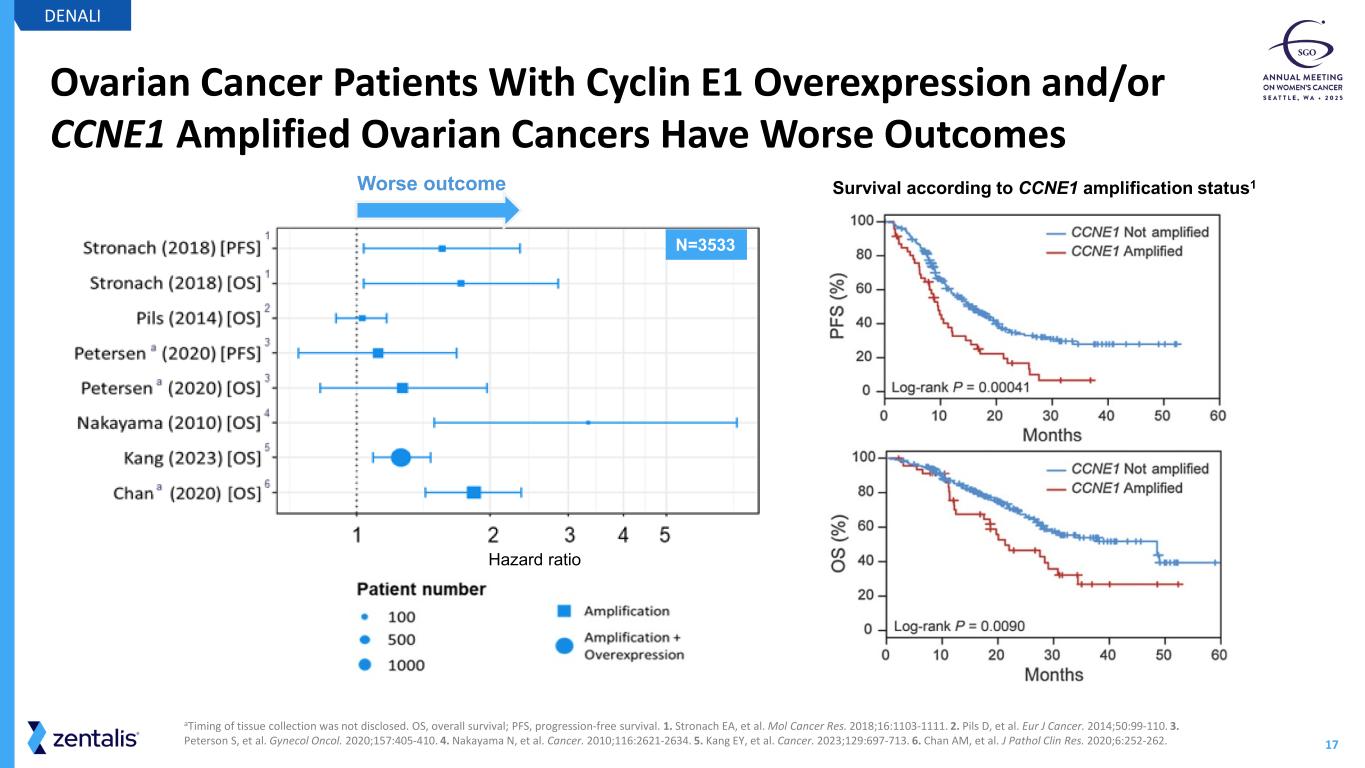
17 Ovarian Cancer Patients With Cyclin E1 Overexpression and/or CCNE1 Amplified Ovarian Cancers Have Worse Outcomes DENALI Survival according to CCNE1 amplification status1 aTiming of tissue collection was not disclosed. OS, overall survival; PFS, progression-free survival. 1. Stronach EA, et al. Mol Cancer Res. 2018;16:1103-1111. 2. Pils D, et al. Eur J Cancer. 2014;50:99-110. 3. Peterson S, et al. Gynecol Oncol. 2020;157:405-410. 4. Nakayama N, et al. Cancer. 2010;116:2621-2634. 5. Kang EY, et al. Cancer. 2023;129:697-713. 6. Chan AM, et al. J Pathol Clin Res. 2020;6:252-262. Hazard ratio N=3533 Worse outcome

18 Study Design Platinum-resistant ovarian cancer 1-5 prior lines of therapy Tissue mandatory for biomarker assessment Key Eligibility DENALI Part 1b Evaluated 400mg 5:2 QD and Confirmed Cyclin E1 Biomarker DENALI Abbreviations: QD, once daily; 5:2, 5-days of treatment followed by 2-days off treatment; ORR, objective response rate; DOR, duration of response; PFS, progression-free survival Endpoints PFS Safety and Tolerability ORR, DOR Enrollment (N=102) Azenosertib monotherapy 400 mg QD 5:2 Part 1b Enrollment CompleteStatus
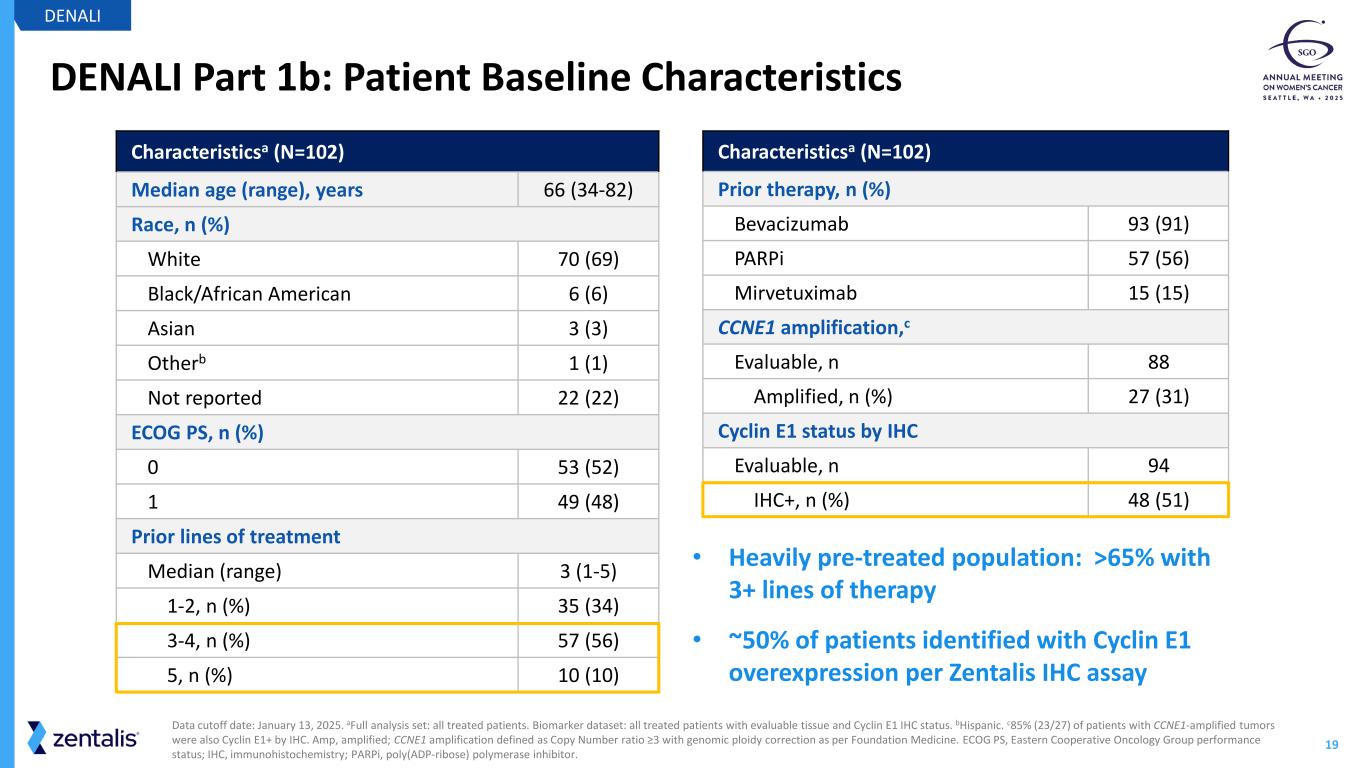
19 DENALI Part 1b: Patient Baseline Characteristics DENALI Data cutoff date: January 13, 2025. aFull analysis set: all treated patients. Biomarker dataset: all treated patients with evaluable tissue and Cyclin E1 IHC status. bHispanic. c85% (23/27) of patients with CCNE1-amplified tumors were also Cyclin E1+ by IHC. Amp, amplified; CCNE1 amplification defined as Copy Number ratio ≥3 with genomic ploidy correction as per Foundation Medicine. ECOG PS, Eastern Cooperative Oncology Group performance status; IHC, immunohistochemistry; PARPi, poly(ADP-ribose) polymerase inhibitor. Characteristicsa (N=102) Median age (range), years 66 (34-82) Race, n (%) White 70 (69) Black/African American 6 (6) Asian 3 (3) Otherb 1 (1) Not reported 22 (22) ECOG PS, n (%) 0 53 (52) 1 49 (48) Prior lines of treatment Median (range) 3 (1-5) 1-2, n (%) 35 (34) 3-4, n (%) 57 (56) 5, n (%) 10 (10) Characteristicsa (N=102) Prior therapy, n (%) Bevacizumab 93 (91) PARPi 57 (56) Mirvetuximab 15 (15) CCNE1 amplification,c Evaluable, n 88 Amplified, n (%) 27 (31) Cyclin E1 status by IHC Evaluable, n 94 IHC+, n (%) 48 (51) • Heavily pre-treated population: >65% with 3+ lines of therapy • ~50% of patients identified with Cyclin E1 overexpression per Zentalis IHC assay
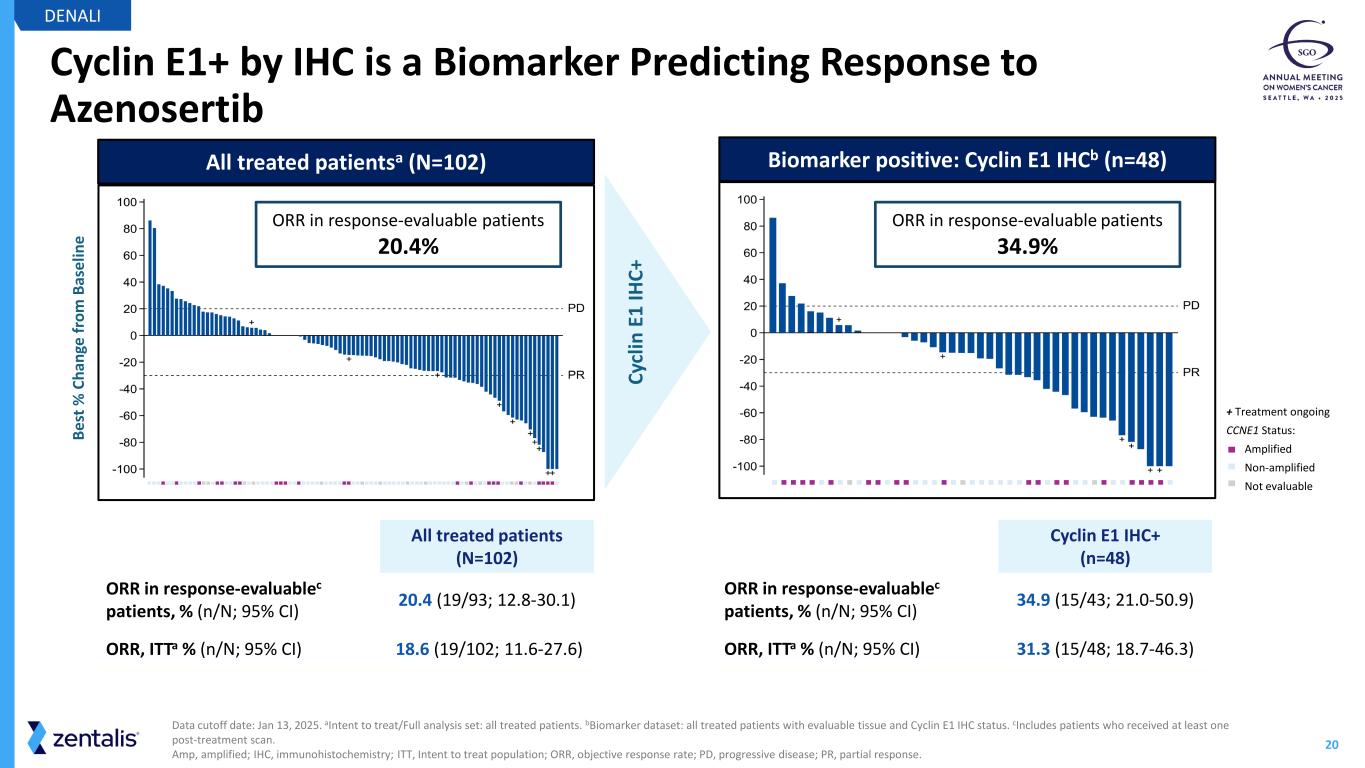
20 Cyclin E1+ by IHC is a Biomarker Predicting Response to Azenosertib DENALI Data cutoff date: Jan 13, 2025. aIntent to treat/Full analysis set: all treated patients. bBiomarker dataset: all treated patients with evaluable tissue and Cyclin E1 IHC status. cIncludes patients who received at least one post-treatment scan. Amp, amplified; IHC, immunohistochemistry; ITT, Intent to treat population; ORR, objective response rate; PD, progressive disease; PR, partial response. All treated patients (N=102) ORR in response-evaluablec patients, % (n/N; 95% CI) 20.4 (19/93; 12.8-30.1) ORR, ITTa % (n/N; 95% CI) 18.6 (19/102; 11.6-27.6) Cyclin E1 IHC+ (n=48) ORR in response-evaluablec patients, % (n/N; 95% CI) 34.9 (15/43; 21.0-50.9) ORR, ITTa % (n/N; 95% CI) 31.3 (15/48; 18.7-46.3) All treated patientsa (N=102) Cy cl in E 1 IH C+ Biomarker positive: Cyclin E1 IHCb (n=48) ORR in response-evaluable patients 20.4% ORR in response-evaluable patients 34.9% + Treatment ongoing CCNE1 Status: Amplified Non-amplified Not evaluable Be st % C ha ng e fr om B as el in e
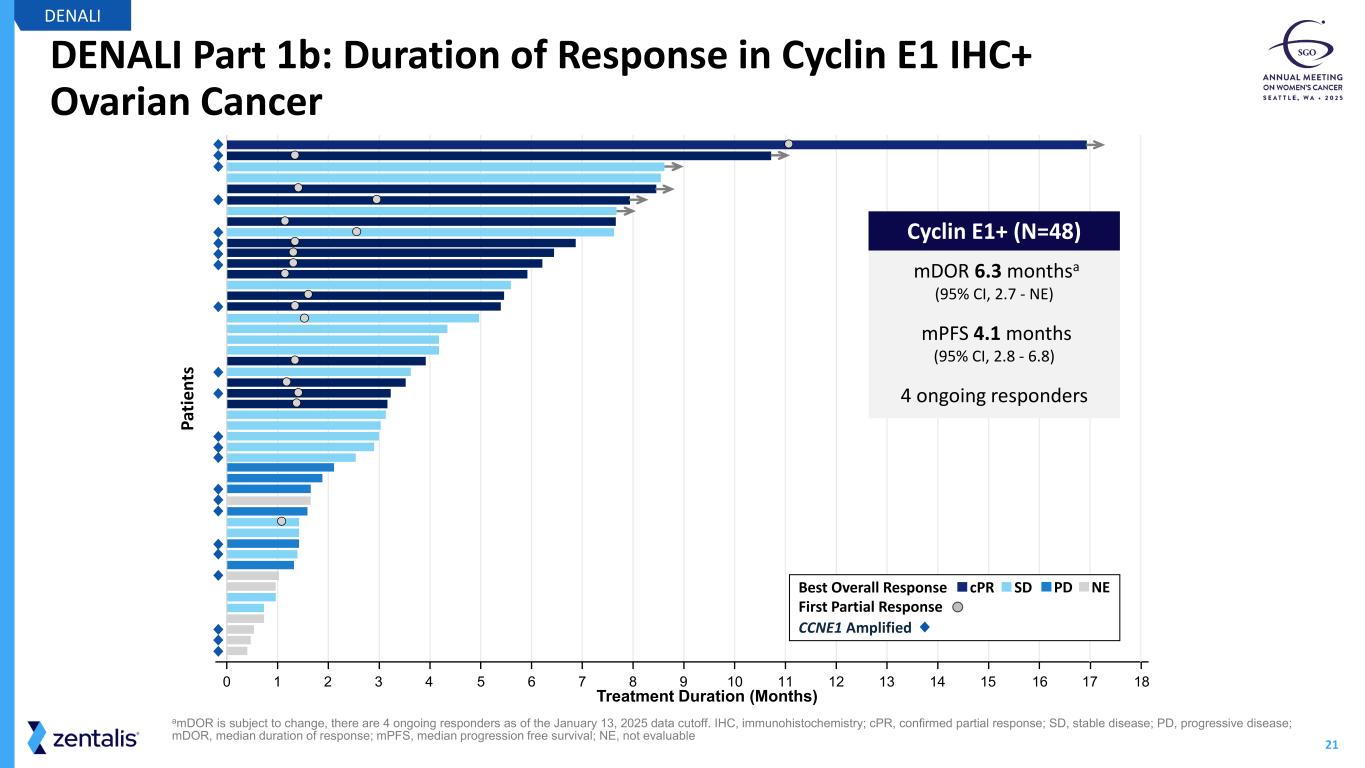
21 DENALI Part 1b: Duration of Response in Cyclin E1 IHC+ Ovarian Cancer DENALI amDOR is subject to change, there are 4 ongoing responders as of the January 13, 2025 data cutoff. IHC, immunohistochemistry; cPR, confirmed partial response; SD, stable disease; PD, progressive disease; mDOR, median duration of response; mPFS, median progression free survival; NE, not evaluable 17 180 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 Pa tie nt s Treatment Duration (Months) mDOR 6.3 monthsa (95% CI, 2.7 - NE) mPFS 4.1 months (95% CI, 2.8 - 6.8) 4 ongoing responders Cyclin E1+ (N=48) PDSDcPRBest Overall Response First Partial Response CCNE1 Amplified NE
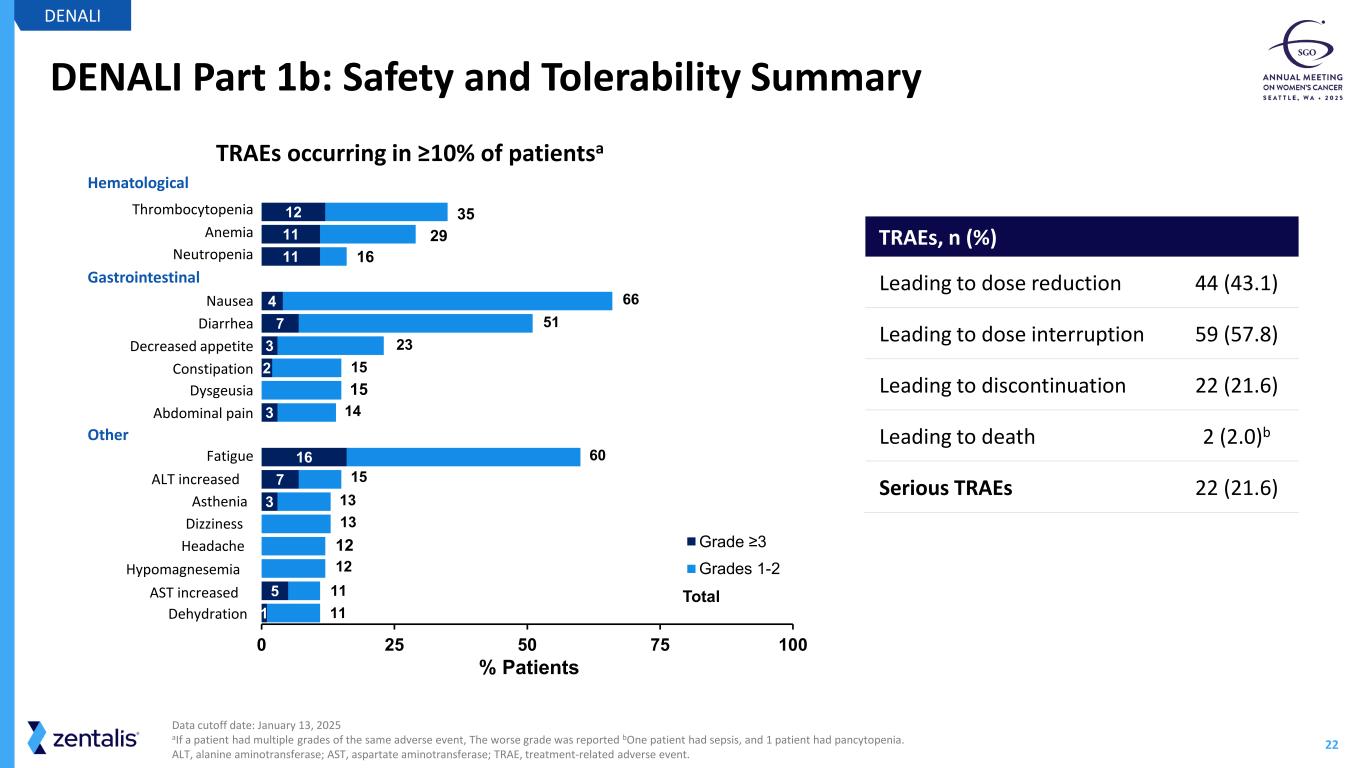
22 DENALI Part 1b: Safety and Tolerability Summary DENALI Data cutoff date: January 13, 2025 aIf a patient had multiple grades of the same adverse event, The worse grade was reported bOne patient had sepsis, and 1 patient had pancytopenia. ALT, alanine aminotransferase; AST, aspartate aminotransferase; TRAE, treatment-related adverse event. 12 11 11 4 7 3 2 3 16 7 3 5 1 0 25 50 75 100 Grade ≥3 Grades 1-2 66 51 15 14 60 15 13 13 12 11 11 12 Total 15 23 % Patients Hematological Thrombocytopenia Anemia Neutropenia Gastrointestinal Nausea Diarrhea Decreased appetite Constipation Dysgeusia Abdominal pain Other Fatigue ALT increased Asthenia Dizziness Headache Hypomagnesemia AST increased Dehydration TRAEs, n (%) Leading to dose reduction 44 (43.1) Leading to dose interruption 59 (57.8) Leading to discontinuation 22 (21.6) Leading to death 2 (2.0)b Serious TRAEs 22 (21.6) 35 29 16 TRAEs occurring in ≥10% of patientsa

23 Registration-Intent DENALI Part 2 Topline Data Anticipated by YE2026 Cyclin E1+ / overexpression by IHC represents ~ 50%c of PROC • Cyclin E1+ IHC more than doubles the eligible patient population beyond CCNE1 amp Azenosertib demonstrates ORR of ~35% in evaluablea patients with Cyclin E1+ PROC • Median duration of response is 6.3 monthsb Cyclin E1 by IHC is a predictive biomarker for response to azenosertib Manageable safety profile • GI toxicity (nausea, diarrhea) and fatigue are the most common TRAEs; although less common, hematological toxicities require close monitoring during treatment with azenosertib Azenosertib data support further development in Cyclin E1+ PROC in Part 2 of the ongoing DENALI study DENALI aIncludes patients who received at least one post treatment scan as of the January 13, 2025 data cut off; bmDOR is subject to change, there are 4 ongoing responders as of the January 13, 2025 data cut off; c Cyclin E1 IHC+ based on Zentalis proprietary IHC cutoff and Cyclin E1 IHC assay developed from the existing clinical data; Cyclin E1 IHC+% based on literature (~20% CCNE1 amplification) and the unbiased CCNE1 amp & Cyclin E1 overlapping data generated from Zentalis clinical trial samples GI, gastrointestinal; mDOR, median duration of response; PROC, platinum-resistant ovarian cancer; TRAE, treatment-related adverse event.

ZN-c3-001 Dose-Escalating Monotherapy Study in Solid Tumors NCT04158336
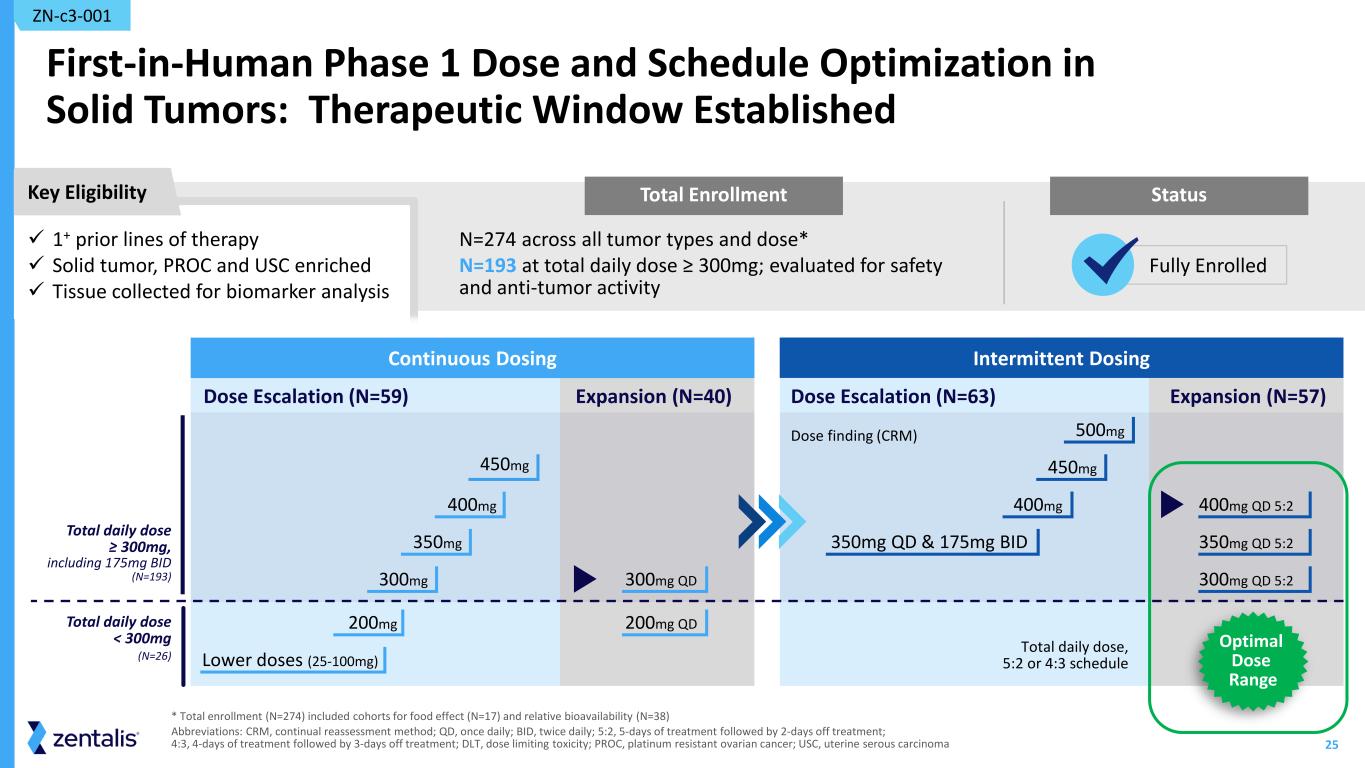
25 Total Enrollment First-in-Human Phase 1 Dose and Schedule Optimization in Solid Tumors: Therapeutic Window Established N=274 across all tumor types and dose* N=193 at total daily dose ≥ 300mg; evaluated for safety and anti-tumor activity ZN-c3-001 * Total enrollment (N=274) included cohorts for food effect (N=17) and relative bioavailability (N=38) Abbreviations: CRM, continual reassessment method; QD, once daily; BID, twice daily; 5:2, 5-days of treatment followed by 2-days off treatment; 4:3, 4-days of treatment followed by 3-days off treatment; DLT, dose limiting toxicity; PROC, platinum resistant ovarian cancer; USC, uterine serous carcinoma Intermittent Dosing Dose finding (CRM) Expansion (N=57)Dose Escalation (N=63) 450mg 500mg 400mg 400mg QD 5:2 350mg QD & 175mg BID 350mg QD 5:2 300mg QD 5:2 Total daily dose, 5:2 or 4:3 schedule Continuous Dosing Dose Escalation (N=59) Expansion (N=40) 450mg 200mg 400mg 350mg Lower doses (25-100mg) 300mg 300mg QD 200mg QD Total daily dose ≥ 300mg, including 175mg BID (N=193) Total daily dose < 300mg (N=26) Key Eligibility Status 1+ prior lines of therapy Solid tumor, PROC and USC enriched Tissue collected for biomarker analysis Fully Enrolled Optimal Dose Range
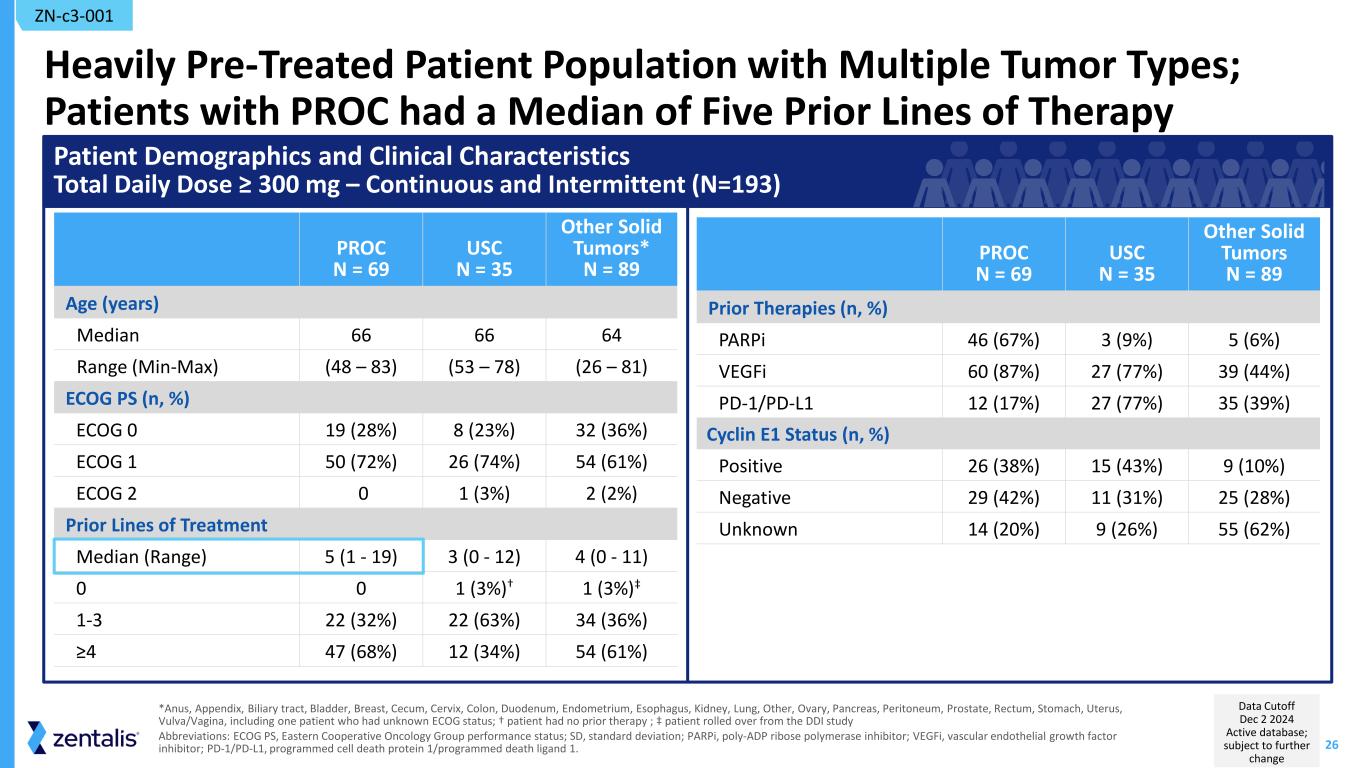
26 Heavily Pre-Treated Patient Population with Multiple Tumor Types; Patients with PROC had a Median of Five Prior Lines of Therapy PROC N = 69 USC N = 35 Other Solid Tumors* N = 89 Age (years) Median 66 66 64 Range (Min-Max) (48 – 83) (53 – 78) (26 – 81) ECOG PS (n, %) ECOG 0 19 (28%) 8 (23%) 32 (36%) ECOG 1 50 (72%) 26 (74%) 54 (61%) ECOG 2 0 1 (3%) 2 (2%) Prior Lines of Treatment Median (Range) 5 (1 - 19) 3 (0 - 12) 4 (0 - 11) 0 0 1 (3%)† 1 (3%)‡ 1-3 22 (32%) 22 (63%) 34 (36%) ≥4 47 (68%) 12 (34%) 54 (61%) *Anus, Appendix, Biliary tract, Bladder, Breast, Cecum, Cervix, Colon, Duodenum, Endometrium, Esophagus, Kidney, Lung, Other, Ovary, Pancreas, Peritoneum, Prostate, Rectum, Stomach, Uterus, Vulva/Vagina, including one patient who had unknown ECOG status; † patient had no prior therapy ; ‡ patient rolled over from the DDI study Abbreviations: ECOG PS, Eastern Cooperative Oncology Group performance status; SD, standard deviation; PARPi, poly-ADP ribose polymerase inhibitor; VEGFi, vascular endothelial growth factor inhibitor; PD-1/PD-L1, programmed cell death protein 1/programmed death ligand 1. Patient Demographics and Clinical Characteristics Total Daily Dose ≥ 300 mg – Continuous and Intermittent (N=193) PROC N = 69 USC N = 35 Other Solid Tumors N = 89 Prior Therapies (n, %) PARPi 46 (67%) 3 (9%) 5 (6%) VEGFi 60 (87%) 27 (77%) 39 (44%) PD-1/PD-L1 12 (17%) 27 (77%) 35 (39%) Cyclin E1 Status (n, %) Positive 26 (38%) 15 (43%) 9 (10%) Negative 29 (42%) 11 (31%) 25 (28%) Unknown 14 (20%) 9 (26%) 55 (62%) ZN-c3-001 Data Cutoff Dec 2 2024 Active database; subject to further change
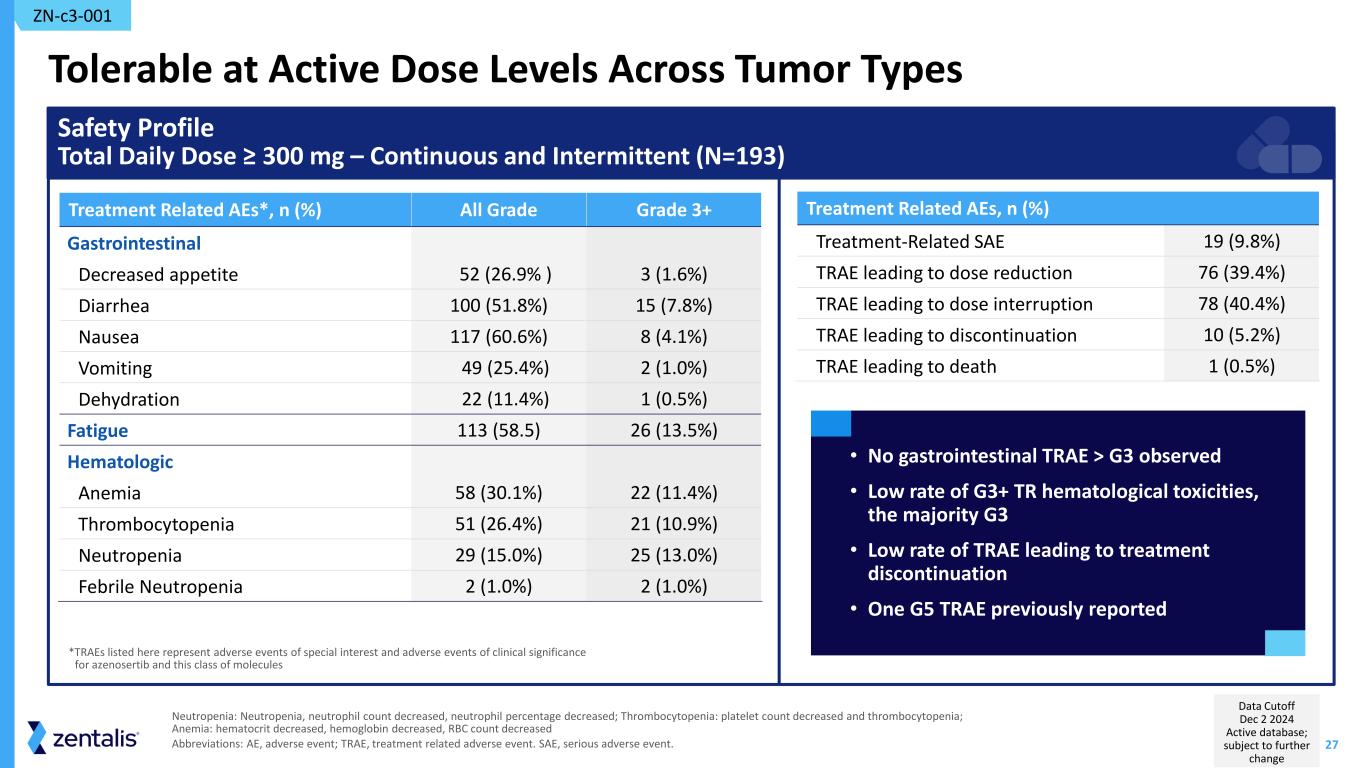
27 Tolerable at Active Dose Levels Across Tumor Types Treatment Related AEs, n (%) Treatment-Related SAE 19 (9.8%) TRAE leading to dose reduction 76 (39.4%) TRAE leading to dose interruption 78 (40.4%) TRAE leading to discontinuation 10 (5.2%) TRAE leading to death 1 (0.5%) Treatment Related AEs*, n (%) All Grade Grade 3+ Gastrointestinal Decreased appetite 52 (26.9% ) 3 (1.6%) Diarrhea 100 (51.8%) 15 (7.8%) Nausea 117 (60.6%) 8 (4.1%) Vomiting 49 (25.4%) 2 (1.0%) Dehydration 22 (11.4%) 1 (0.5%) Fatigue 113 (58.5) 26 (13.5%) Hematologic Anemia 58 (30.1%) 22 (11.4%) Thrombocytopenia 51 (26.4%) 21 (10.9%) Neutropenia 29 (15.0%) 25 (13.0%) Febrile Neutropenia 2 (1.0%) 2 (1.0%) Safety Profile Total Daily Dose ≥ 300 mg – Continuous and Intermittent (N=193) • No gastrointestinal TRAE > G3 observed • Low rate of G3+ TR hematological toxicities, the majority G3 • Low rate of TRAE leading to treatment discontinuation • One G5 TRAE previously reported *TRAEs listed here represent adverse events of special interest and adverse events of clinical significance for azenosertib and this class of molecules ZN-c3-001 Neutropenia: Neutropenia, neutrophil count decreased, neutrophil percentage decreased; Thrombocytopenia: platelet count decreased and thrombocytopenia; Anemia: hematocrit decreased, hemoglobin decreased, RBC count decreased Abbreviations: AE, adverse event; TRAE, treatment related adverse event. SAE, serious adverse event. Data Cutoff Dec 2 2024 Active database; subject to further change
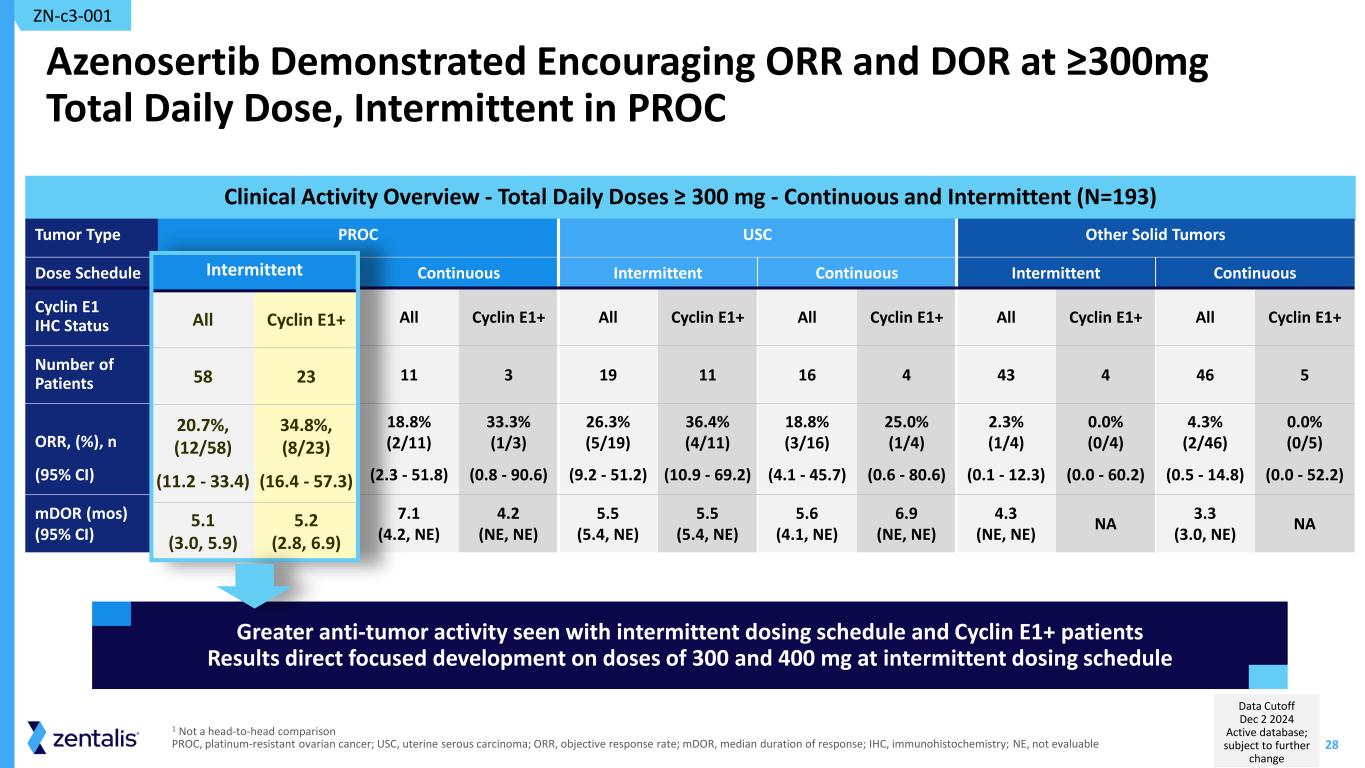
28 Clinical Activity Overview - Total Daily Doses ≥ 300 mg - Continuous and Intermittent (N=193) Azenosertib Demonstrated Encouraging ORR and DOR at ≥300mg Total Daily Dose, Intermittent in PROC Tumor Type PROC USC Other Solid Tumors Dose Schedule Intermittent Continuous Intermittent Continuous Intermittent Continuous Cyclin E1 IHC Status All Cyclin E1+ All Cyclin E1+ All Cyclin E1+ All Cyclin E1+ All Cyclin E1+ All Cyclin E1+ Number of Patients 58 23 11 3 19 11 16 4 43 4 46 5 ORR, (%), n (95% CI) 20.7%, (12/58) (11.2 - 33.4) 34.8%, (8/23) (16.4 - 57.3) 18.8% (2/11) (2.3 - 51.8) 33.3% (1/3) (0.8 - 90.6) 26.3% (5/19) (9.2 - 51.2) 36.4% (4/11) (10.9 - 69.2) 18.8% (3/16) (4.1 - 45.7) 25.0% (1/4) (0.6 - 80.6) 2.3% (1/4) (0.1 - 12.3) 0.0% (0/4) (0.0 - 60.2) 4.3% (2/46) (0.5 - 14.8) 0.0% (0/5) (0.0 - 52.2) mDOR (mos) (95% CI) 5.1 (3.0, 5.9) 5.2 (2.8, 6.9) 7.1 (4.2, NE) 4.2 (NE, NE) 5.5 (5.4, NE) 5.5 (5.4, NE) 5.6 (4.1, NE) 6.9 (NE, NE) 4.3 (NE, NE) NA 3.3 (3.0, NE) NA ZN-c3-001 1 Not a head-to-head comparison PROC, platinum-resistant ovarian cancer; USC, uterine serous carcinoma; ORR, objective response rate; mDOR, median duration of response; IHC, immunohistochemistry; NE, not evaluable Greater anti-tumor activity seen with intermittent dosing schedule and Cyclin E1+ patients Results direct focused development on doses of 300 and 400 mg at intermittent dosing schedule Inter itt t All Cycli 58 20.7 , (12/58) (11.2 - 33.4) 34.8 , (8/23) (16.4 - 57.3) 5.1 (3.0, 5.9) 5.2 (2.8, 6.9) Data Cutoff Dec 2 2024 Active database; subject to further change

29 Key Takeaways from 001 Azenosertib studied in a large patient population across multiple tumor types Platinum-resistant ovarian cancer (PROC) identified as an indication particularly susceptible to WEE1 inhibition Cyclin E1 identified as predictive biomarker for response to azenosertib Meaningful therapeutic window identified providing a favorable risk-benefit profile in Cyclin E1+ PROC patients at total daily doses of 300 and 400mg QD 5:2 ZN-c3-001

MAMMOTH (ZN-c3-006) NCT05198804

31 MAMMOTH: Two Clinically-active Monotherapy Doses Studied in Heavily Pre-treated, PARPi-resistant PROC Patient Population Study Design 1-5 prior lines of therapy Platinum-resistant, progressed while receiving an approved PARP inhibitor Mandatory sufficient tissue for biomarker analysis Key Eligibility MAMMOTH Abbreviations: QD, once daily; 5:2, 5-days of treatment followed by 2-days off treatment; ORR, objective response rate DOR, duration of response; PFS, progression-free survival; PROC, platinum-resistant ovarian cancer; PARPi, poly (ADP-ribose) polymerase inhibitor Azenosertib + niraparib Concurrent schedule (N=28) Azenosertib + niraparib Alternating schedule (N=28) Azenosertib monotherapy 300 or 400 mg QD 5:2 (N=61) PFS Safety and Tolerability ORR, DOR Endpoints Enrollment (N=117) Enrollment CompleteStatus
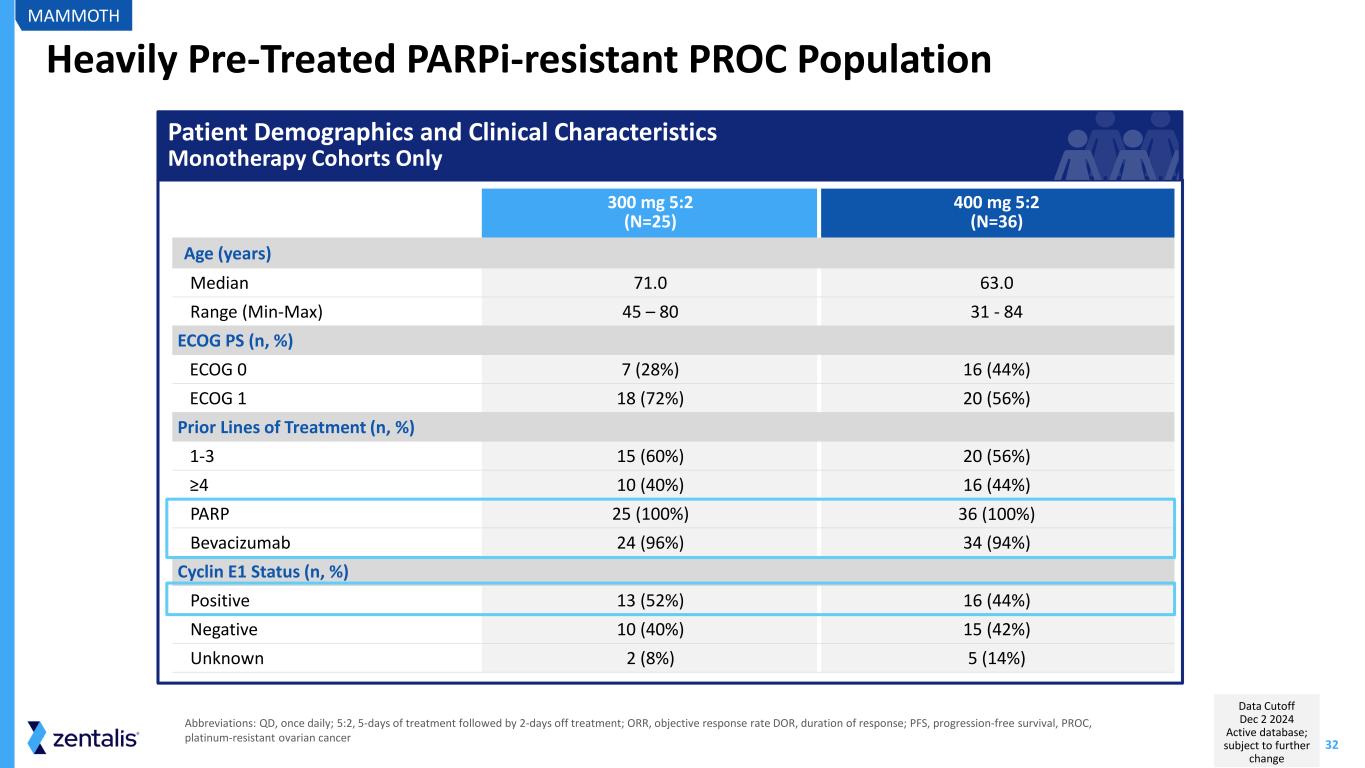
32 Heavily Pre-Treated PARPi-resistant PROC Population 300 mg 5:2 (N=25) 400 mg 5:2 (N=36) Age (years) Median 71.0 63.0 Range (Min-Max) 45 – 80 31 - 84 ECOG PS (n, %) ECOG 0 7 (28%) 16 (44%) ECOG 1 18 (72%) 20 (56%) Prior Lines of Treatment (n, %) 1-3 15 (60%) 20 (56%) ≥4 10 (40%) 16 (44%) PARP 25 (100%) 36 (100%) Bevacizumab 24 (96%) 34 (94%) Cyclin E1 Status (n, %) Positive 13 (52%) 16 (44%) Negative 10 (40%) 15 (42%) Unknown 2 (8%) 5 (14%) MAMMOTH Abbreviations: QD, once daily; 5:2, 5-days of treatment followed by 2-days off treatment; ORR, objective response rate DOR, duration of response; PFS, progression-free survival, PROC, platinum-resistant ovarian cancer Patient Demographics and Clinical Characteristics Monotherapy Cohorts Only Data Cutoff Dec 2 2024 Active database; subject to further change
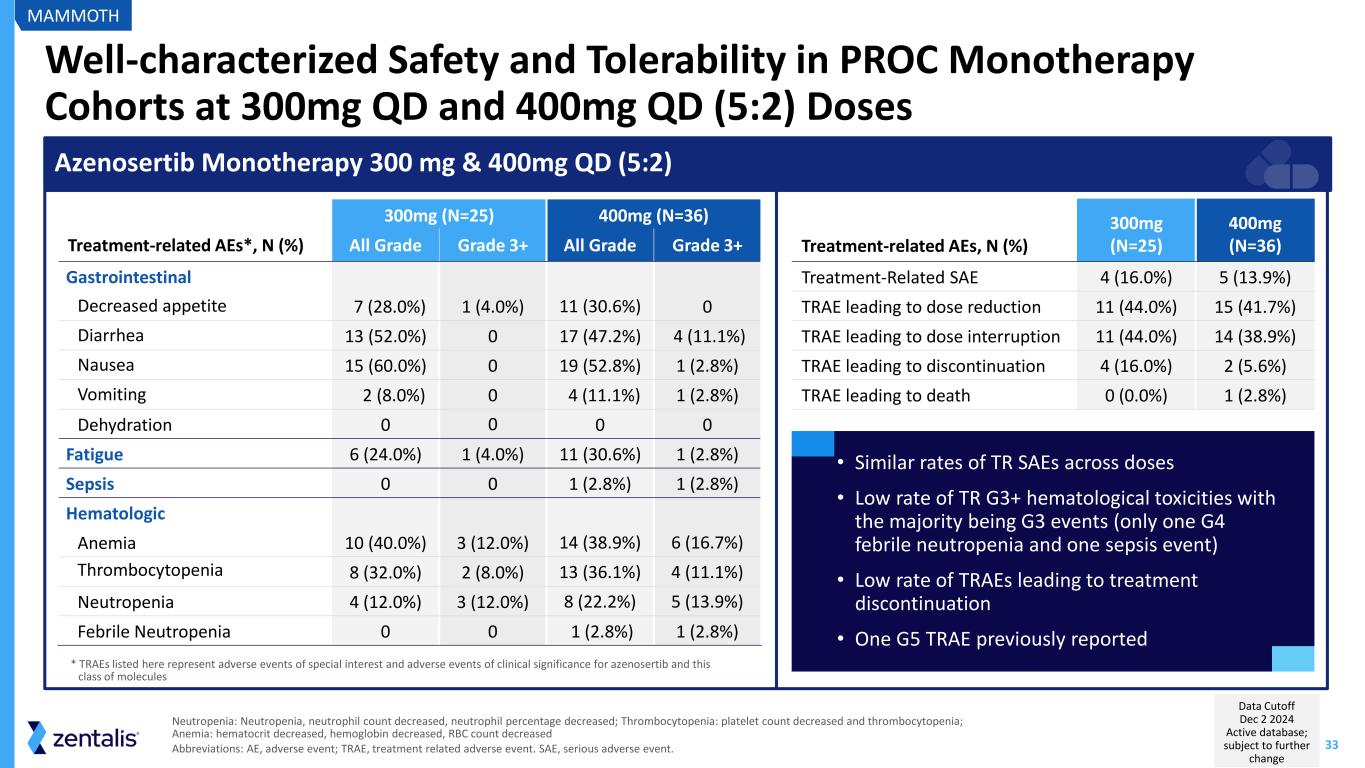
33 Well-characterized Safety and Tolerability in PROC Monotherapy Cohorts at 300mg QD and 400mg QD (5:2) Doses Neutropenia: Neutropenia, neutrophil count decreased, neutrophil percentage decreased; Thrombocytopenia: platelet count decreased and thrombocytopenia; Anemia: hematocrit decreased, hemoglobin decreased, RBC count decreased Abbreviations: AE, adverse event; TRAE, treatment related adverse event. SAE, serious adverse event. 300mg (N=25) 400mg (N=36) Treatment-related AEs*, N (%) All Grade Grade 3+ All Grade Grade 3+ Gastrointestinal Decreased appetite 7 (28.0%) 1 (4.0%) 11 (30.6%) 0 Diarrhea 13 (52.0%) 0 17 (47.2%) 4 (11.1%) Nausea 15 (60.0%) 0 19 (52.8%) 1 (2.8%) Vomiting 2 (8.0%) 0 4 (11.1%) 1 (2.8%) Dehydration 0 0 0 0 Fatigue 6 (24.0%) 1 (4.0%) 11 (30.6%) 1 (2.8%) Sepsis 0 0 1 (2.8%) 1 (2.8%) Hematologic Anemia 10 (40.0%) 3 (12.0%) 14 (38.9%) 6 (16.7%) Thrombocytopenia 8 (32.0%) 2 (8.0%) 13 (36.1%) 4 (11.1%) Neutropenia 4 (12.0%) 3 (12.0%) 8 (22.2%) 5 (13.9%) Febrile Neutropenia 0 0 1 (2.8%) 1 (2.8%) Azenosertib Monotherapy 300 mg & 400mg QD (5:2) Treatment-related AEs, N (%) 300mg (N=25) 400mg (N=36) Treatment-Related SAE 4 (16.0%) 5 (13.9%) TRAE leading to dose reduction 11 (44.0%) 15 (41.7%) TRAE leading to dose interruption 11 (44.0%) 14 (38.9%) TRAE leading to discontinuation 4 (16.0%) 2 (5.6%) TRAE leading to death 0 (0.0%) 1 (2.8%) • Similar rates of TR SAEs across doses • Low rate of TR G3+ hematological toxicities with the majority being G3 events (only one G4 febrile neutropenia and one sepsis event) • Low rate of TRAEs leading to treatment discontinuation • One G5 TRAE previously reported * TRAEs listed here represent adverse events of special interest and adverse events of clinical significance for azenosertib and this class of molecules MAMMOTH Data Cutoff Dec 2 2024 Active database; subject to further change
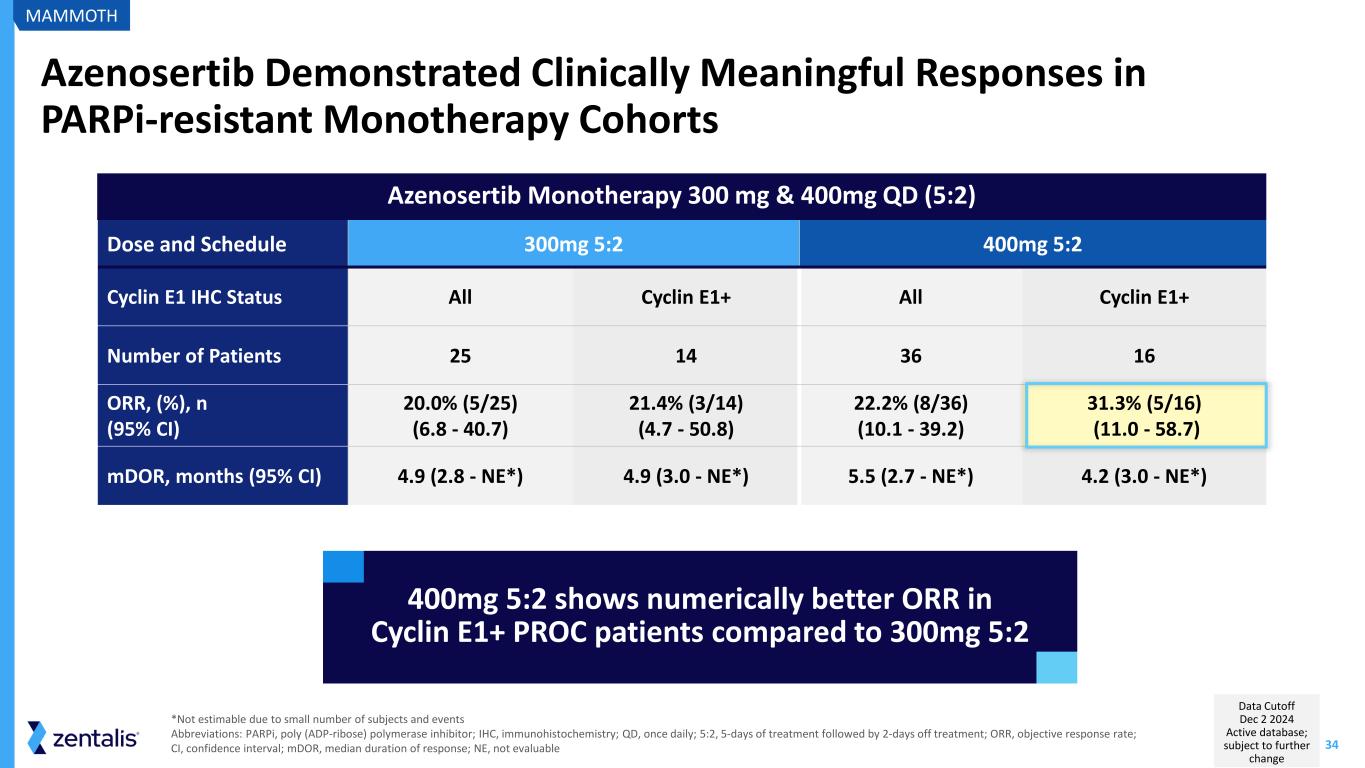
34 Azenosertib Monotherapy 300 mg & 400mg QD (5:2) Azenosertib Demonstrated Clinically Meaningful Responses in PARPi-resistant Monotherapy Cohorts Dose and Schedule 300mg 5:2 400mg 5:2 Cyclin E1 IHC Status All Cyclin E1+ All Cyclin E1+ Number of Patients 25 14 36 16 ORR, (%), n (95% CI) 20.0% (5/25) (6.8 - 40.7) 21.4% (3/14) (4.7 - 50.8) 22.2% (8/36) (10.1 - 39.2) 31.3% (5/16) (11.0 - 58.7) mDOR, months (95% CI) 4.9 (2.8 - NE*) 4.9 (3.0 - NE*) 5.5 (2.7 - NE*) 4.2 (3.0 - NE*) 400mg 5:2 shows numerically better ORR in Cyclin E1+ PROC patients compared to 300mg 5:2 *Not estimable due to small number of subjects and events Abbreviations: PARPi, poly (ADP-ribose) polymerase inhibitor; IHC, immunohistochemistry; QD, once daily; 5:2, 5-days of treatment followed by 2-days off treatment; ORR, objective response rate; CI, confidence interval; mDOR, median duration of response; NE, not evaluable MAMMOTH Data Cutoff Dec 2 2024 Active database; subject to further change

35 Key Takeaways from MAMMOTH Monotherapy Cohort Consistent antitumor activity in PARPi-resistant patients Tolerability and toxicity consistent with 001 study and similar between the assessed doses 400mg QD 5:2 showed numerically higher response rates than 300mg QD 5:2 MAMMOTH Learnings continue to support development of azenosertib in Cyclin E1+ PROC Abbreviations: QD, once daily; 5:2, 5-days of treatment followed by 2-days off treatment; PROC, platinum-resistant ovarian cancer; PARPi, poly (ADP-ribose) polymerase inhibitor

36 INDICATION TRIAL NAME + DEVELOPMENT APPROACH PHASE 1 PHASE 2 PHASE 3 MILESTONE* Studies for Lead Indication PROC Monotherapy DENALI Part 1b DENALI Part 2a + Part 2b Registration-intent Cyclin E1-Positive FDA Fast Track Designation Topline Data YE2026 Randomized Confirmatory Study Azenosertib vs. SOC chemo Cyclin E1-Positive Initiation 2026 Other Studies Ongoing Uterine Serous Carcinoma Monotherapy TETON (ZN-c3-004) FDA Fast Track Designation Data 1H26 Ovarian Cancer Combination ZN-c3-002 Azenosertib + multiple chemo backbones and bevacizumab Currently Enrolling Bevacizumab Combo Ongoing Study Planned Study*Anticipated Azenosertib has the Potential to be a First-in-Class and Best-in-Class WEE1 Inhibitor for Patients with Ovarian Cancer and Other Tumor Types

zentalis.com Science Center 10275 Science Center Drive Suite 200 San Diego, CA 92121 Julie Eastland Chief Executive Officer jeastland@zentalis.com Haibo Wang Chief Business Officer hwang@zentalis.com Aron Feingold VP, IR and Corporate Communications afeingold@zentalis.com
